The relationship between combined oral contraceptive (COC) use and breast cancer risk is complex and has generated both research and vigorous discussion. Although the increase in breast cancer risk in patients who use COCs is well documented, its interpretation in clinical practice requires context. Most notably, we must know what we are comparing this increased risk with. Unless we understand baseline risk and how the reported increases are evaluated, we lack perspective for counseling individual patients.
Takeaways
- Breast cancer risk increases minimally with ethinyl estradiol–containing oral contraceptive use, is observed during the period of use, and returns to baseline within a few years of cessation of use.
- The risk increase seen in studies is primarily over a follow-up of 10 years or less, meaning any measured increase relates to an excess in 10-year risk.
- The increase in breast cancer risk while using ethinyl estradiol–containing oral contraceptives is similar to the increase observed following a pregnancy with delivery, which increases breast cancer risk by a similar amount, remains elevated for about 10 years, and abates thereafter.
- For any patient 40 years or younger, the approximate risk of breast cancer while using ethinyl estradiol–containing oral contraceptives is still lower than the risk at which routine mammography is recommended in national screening guidelines.
- Any use of ethinyl estradiol–containing oral contraceptives does not increase lifetime breast cancer risk or the risk of death from breast cancer.
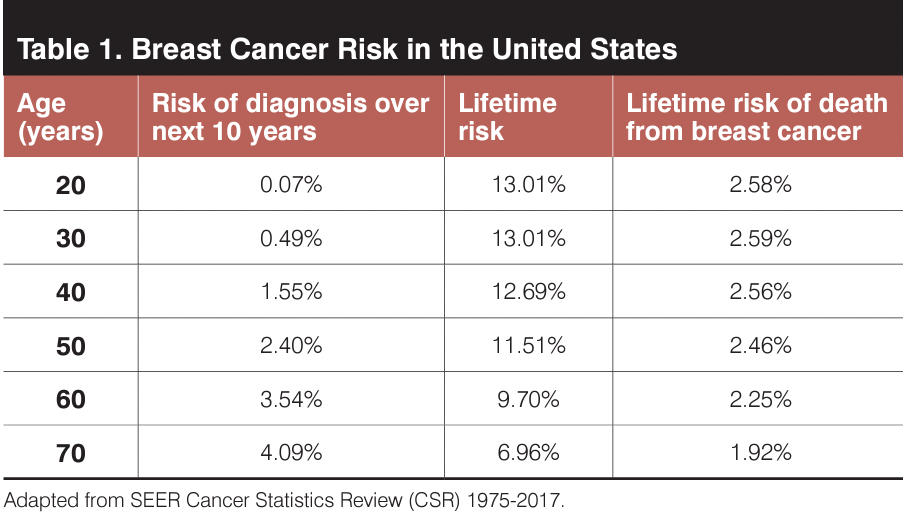
Oral contraceptives are the most used reversible method of contraception in the United States.1 Most women who use oral contraceptive are 29 years or younger, with approximately 33% of them aged 15 to 19 years and 37% aged 20 to 29 years. Breast cancer is rare in young people with breast tissue who are genetically female, especially those under 30 years of age (Table 1). Breast cancer risk rises with age, from a rate of 10.5/100,000 at age 25 to 29 to 133/100,000 annually at age 40 to 44 years. The known risk factors for the premenopausal population include family history and genetic susceptibility, a finding of atypical hyperplasia on breast biopsy, parity past age 30 years, and a sedentary lifestyle.2,3 These are not always controlled for adequately in cohort studies that evaluate risk.
Early studies of combined oral contraceptives and breast cancer risk
Data from early studies reported in the 1980s included evaluations from large cohorts that reflected COC use in prior decades, including the Royal College of General Practitioners, Oxford-Family Planning Association, Walnut Creek Contraceptive Drug Study, and Nurses’ Health Study.4-8 These studies included participants who were primarily married couples spacing births, and the data showed no difference in breast cancer rates between those who took COCs and those who did not.
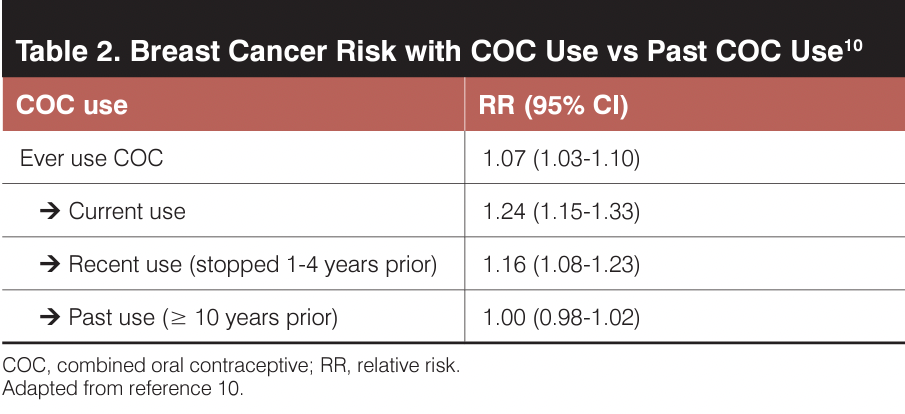
COC use changed in the 1980s compared with that observed in earlier cohort studies, as individuals tended to be younger and took oral contraceptives to delay initial pregnancy and for longer durations.9 Insights into the changing demographics of those taking oral contraceptives was reflected in publications in the 1990s from the Collaborative Group on Hormonal Factors in Breast Cancer.10 These authors reanalyzed data from 54 studies from 26 countries, which included 53,297 participants with breast cancer and 100,239 without breast cancer.
Of note, these trials primarily included COCs with ethinyl estradiol (EE) doses higher than 30 mcg daily. The authors reported no dose effect among various EE doses when evaluated at less than 50 mcg, 50 mcg, and more than 50 mcg. The analyses demonstrated an increase in breast cancer risk in patients who were currently taking or had recently taken COCs, but no increase in patients who had taken COCs 10 or more years prior (Table 2).10 The authors found no differences in metastatic breast cancer rates between those who took COCs and those who did not.
Although the Collaborative Group evaluation included patients of all ages, some as young as 16 years, Kumle et al11 focused on those who were 30 to 49 years of age. This prospective cohort study of Norwegian and Swedish participants recruited patients in 1991 and 1992 with follow-up through 1999. The authors evaluated 103,027 participants aged 30 to 49 years with contraceptive use information and reported a relative risk in this specific age group of 1.5 (95% CI, 1.0-2.0) among those who took COCs and 1.6 (95% CI 1.0-2.4) among those who took progestin-only pills.
Studies of breast cancer risk in patients taking contemporary COCs
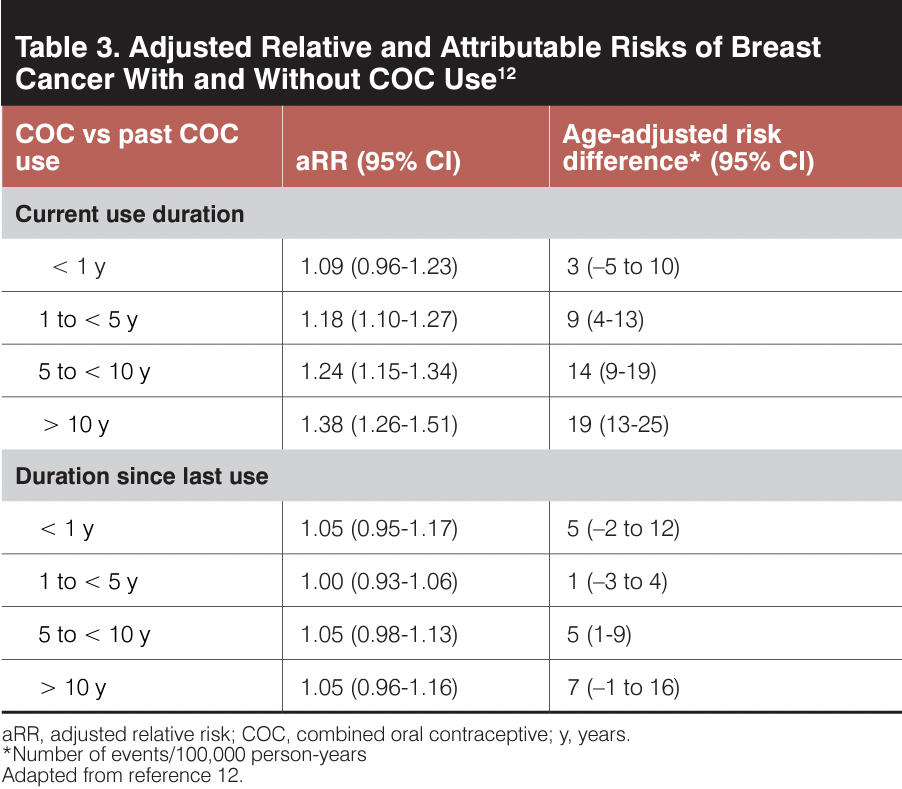
By the late 2010s, more data became available assessing breast cancer risk in large cohorts who had taken contemporary low-dose pills. Mørch et al12 used data from a Danish national registry of patients aged 15 to 49 years who had been prospectively recruited and followed. The authors used multiple available national registries to correlate breast cancer risk and outcomes based on contraceptive use or nonuse. The entire cohort followed 1.8 million Danish women aged 15 to 79 years beginning in January 1995 of whom the authors evaluated those aged 15 to 49 years at entry into the database. Participants were followed until they had a diagnosis of breast cancer, died, emigrated, reached 50 years of age, or until the study period ended (December 31, 2012). The average follow-up of patients in the cohort was 10.9 years plus or minus 5.8 years; the authors did not provide a range.
Overall, 40.1% of the population had never used a hormonal method, 22.3% had used a hormonal method more than 6 months previously, and 37.5% currently or had recently (<6 months) used a hormonal contraceptive (32.3% being a low dose [<50 mcg EE] combined oral contraceptive). Ages among cohorts varied significantly, reflecting contraceptive prescribing patterns in Denmark, with a mean age of 26 to 28 years among those taking various low-dose COCs, 36 years for those who had never taken or who had previously taken hormonal contraceptives, and 40 years for those using levonorgestrel intrauterine devices. Overall, breast cancer risk increased with hormonal contraceptive use (HR, 1.20; 95% CI, 1.14-1.26), with that risk primarily reflected by the largest cohort of patients, those taking low-dose COCs (HR, 1.19; 95% CI, 1.13-1.26), regardless of progestin type. The authors reported an increase in adjusted relative risk of breast cancer with longer duration of COC use, relative to non-COC use, but no increase in risk once COC use stopped (Table 3).12 The estimated number of excess breast cancers per 100,000 individuals each year, even in the group with the largest association (COC use >10 years), was approximately 19/100,000 person-years.
What does increase in breast cancer risk with COC use mean?
How do we use available information to provide accurate counseling for our patients? We need to appreciate 3 important concepts to understand what an “increase in breast cancer risk with combined oral contraceptive use” means. First, the studies mentioned previously, along with others in the literature, have consistently provided similar outcomes. Breast cancer risk, whether assessed as a relative risk or hazard ratio, is increased by 1.2- to 1.5-fold when comparing those who currently take COCs with those who use nonhormonal contraception, with findings from most studies showing risk increases up to 1.3-fold.10-13
A recent meta-analysis confirmed this increase (OR, 1.23; 95% CI, 1.14-1.32).14 Second, the studies evaluating risk generally report outcomes during about 10 years of follow-up.11,12,14 Thus, the risk these studies are describing is an increase in 10-year risk. Third, any increase in risk dissipates with time since last use, with data from older studies suggesting that risk is gone within 10 years after discontinuation and data from more contemporary studies finding that risk may go away even faster.10,12 Evidence from long-term studies confirms that patients who have previously taken COCs have no increase in breast cancer diagnosis, mortality, or all-cause mortality.15-19 The risks described mirror exactly the outcomes seen with pregnancy, in which the risk of breast cancer increases for approximately 10 years after a delivery and then abates thereafter.20,21
Patients and clinicians historically have a poor comprehension of what an increase in medical risk truly means.22 For breast cancer in young patients, we can translate this information realistically based on the information in Table 1. Breast cancer risk increases with age and is low at baseline for average-risk patients under 40 years of age. Even if the 10-year relative risk is estimated to increase by 30% with COC use, the absolute risk for a 20-year-old who takes COCs for 10 years increases from 0.07% to 0.09% and for a 30-year-old from 0.49% to 0.64%. Thus, clinicians can explain to patients aged 20 to 29 years that the increase is negligible because the baseline risk is extremely low, and to those aged 30 to 39 years that the increase is still small, with an absolute risk that is still less than half that for patients aged 40 to 49 years. Another perspective to consider for a patient of average risk is that routine mammographic screening for breast cancer is recommended beginning at 40 years of age, which is when the baseline age-related risk in the United States is 1.55%; COC use for patients younger than 40 years is not increased above this risk rate.
For the average patient aged 40 to 49 years who takes a COC, the 10-year risk with use increases from 1.55% to 2.02%, which is still approximately 20% lower than her age-related risk at 50 years old (2.40%). Overall, clinicians can provide clear information to patients that any breast cancer risk increase is small regardless of age, applies mainly during the period of use, and will return to their baseline within a few years of cessation of use.
For those at higher risk, such as patients with breast cancer gene mutations or a strong family history of disease, clinicians should individualize recommendations because COC use is likely to increase their risk by the same proportion as for those at average risk (ie, a relative increase of 20% to 30%). Data from recent large cohort studies in this population have clarified the associated risks, summarized in a recent meta-analysis.23 Whereas the lifetime risk of ovarian cancer is just over 1% for the average patient, it is 20% to 40% for those who carry the BRCA1/2 genes.24,25 As for average-risk patients, it is helpful to communicate the magnitude of risk in absolute rather than relative terms. For example, the 10-year risk of developing breast cancer for a 38-year-old patient with increased risk due to a combination of reproductive and familial factors may be 5% or higher. Assuming a relative risk of 1.3 with COC use, the absolute 10-year risk estimate for this high-risk patient would be 6.5%. Patients with BRCA1 will see their absolute risk rise from 20% over 10 years25 to 26% over the same period.
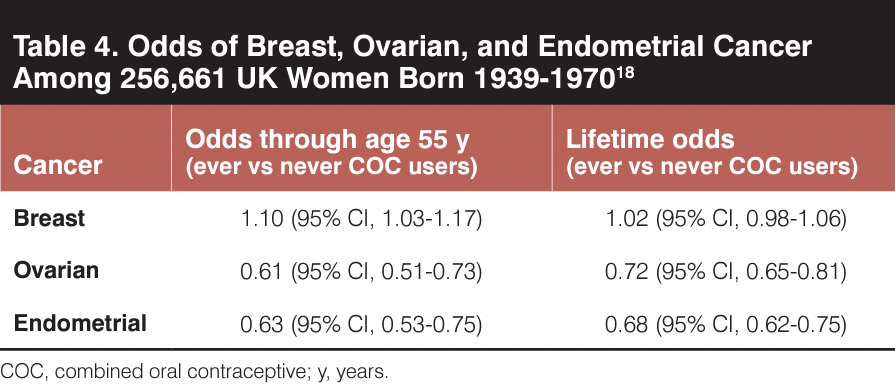
For the average person considering COC use, the translation of these data into clinical advice has lacked clarity and specificity regarding the magnitude of risk; this has resulted in unnecessary fear, especially for patients who are at average risk of breast cancer. A recent UK biobank study evaluated the lifetime risk of breast, ovarian, and endometrial cancer among 256,661 women who were born between 1939 and 1970 and followed through 2019 (Table 4).18 Although the risk of breast cancer appears to be slightly increased through the age of 55 years, lifetime risk is not altered. Comparatively, ovarian and endometrial cancer risks significantly decrease. Importantly, in patients at high risk of breast cancer, ovarian cancer is significantly decreased with COC use.26-29 Balancing these factors for individual patients who are at increased risk for either of these cancers requires nuanced discussion to determine whether any short-term initial increase in breast cancer risk is sufficiently offset by the overall decrease in lifetime ovarian cancer risk.
Future considerations
When data from the Mørch et al12 study were published in 2017, leading authorities pointed out that the findings were nothing new, attempting to reassure clinicians and patients that we have known all along about this increase in risk in patients who take combined hormonal contraceptives with EE-containing contraceptives.30 Importantly, calls for COC formulations that do not increase this risk, even in the short term, have been continuing for decades.31,32 We are entering a new era with estradiol- and estetrol-containing COCs. Whereas estradiol, like EE, stimulates the breast, estetrol is antagonistic at the breast estrogen receptor-alpha membrane receptor.33 This antagonism results in a tamoxifen-like effect in human breast cancer cell lines and an increase in apoptosis in data from clinical studies of patients with advanced breast cancer.34,35
Conclusion
Currently, we can be reassured that any breast cancer risk increase with use of EE-containing oral contraceptives is minimal, relates primarily to excess 10-year risk, and abates after discontinuing use. The next generation of studies will need to address whether COCs containing estetrol decrease breast cancer risk for all cancers, especially with prolonged use.
References
- Daniels K, Abma JC. Current Contraceptive Status Among Women Aged 15-49: United States, 2017-2019. NCHS Data Brief. 2020;(388):1-8.
- Daly AA, Rolph R, Cutress RI, Copson ER. A review of modifiable risk factors in young women for the prevention of breast cancer. Breast Cancer (Dove Med Press). 2021;13:241-57. doi:10.2147/BCTT.S268401
- What Are the Risk Factors for Breast Cancer? U.S. Centers for Disease Control and Prevention. July 25, 2023. Accessed January 3, 2024. https://www.cdc.gov/cancer/breast/basic_info/risk_factors.htm
- Further analyses of mortality in oral contraceptive users. Royal College of General Practitioners' Oral Contraception Study. Lancet. 1981;1(8219):541-546.
- Vessey MP, McPherson K, Yeates D, Doll R. Oral contraceptive use and abortion before first term pregnancy in relation to breast cancer risk. Br J Cancer. 1982;45(3):327-331. doi:10.1038/bjc.1982.58
- Ramcharan S, Pellegrin FA, Ray RM, Hsu JP. The Walnut Creek Contraceptive Drug Study. A prospective study of the side effects of oral contraceptives. Volume III, an interim report: A comparison of disease occurrence leading to hospitalization or death in users and nonusers of oral contraceptives. J Reprod Med. 1980;25(6 Suppl):345-372.
- Vessey MP, McPherson K, Villard-Mackintosh L, Yeates D. Oral contraceptives and breast cancer: latest findings in a large cohort study. Br J Cancer. 1989;59(4):613-7. doi:10.1038/bjc.1989.124
- Romieu I, Willett WC, Colditz GA, Stampfer MJ, Rosner B, Hennekens CH, et al. Prospective study of oral contraceptive use and risk of breast cancer in women. J Natl Cancer Inst. 1989;81(17):1313-21. doi:10.1093/jnci/81.17.1313
- Dawson DA. Trends in use of oral contraceptives--data from the 1987 National Health Interview Survey. Fam Plann Perspect. 1990;22(4):169-72.
- Collaborative Group on Hormonal Factors in Breast Cancer. Breast cancer and hormonal contraceptives: collaborative reanalysis of individual data on 53 297 women with breast cancer and 100 239 women without breast cancer from 54 epidemiological studies. Lancet. 1996;347(9017):1713-27. doi:10.1016/s0140-6736(96)90806-5
- Kumle M, Weiderpass E, Braaten T, Persson I, Adami HO, Lund E. Use of oral contraceptives and breast cancer risk: The Norwegian-Swedish Women's Lifestyle and Health Cohort Study. Cancer Epidemiol Biomarkers Prev. 2002;11(11):1375-81
- Mørch LS, Skovlund CW, Hannaford PC, Iversen L, Fielding S, Lidegaard Ø. Contemporary Hormonal Contraception and the Risk of Breast Cancer. N Engl J Med. 2017;377(23):2228-39. doi:10.1056/NEJMoa1700732
- Burchardt NA, Eliassen AH, Shafrir AL, Rosner B, Tamimi RM, Kaaks R, et al. Oral contraceptive use by formulation and breast cancer risk by subtype in the Nurses' Health Study II: a prospective cohort study. Am J Obstet Gynecol. 2022 Jun;226(6):821.e1-821.e26. doi:10.1016/j.ajog.2021.12.022
- Fitzpatrick D, Pirie K, Reeves G, Green J, Beral V. Combined and progestagen-only hormonal contraceptives and breast cancer risk: A UK nested case-control study and meta-analysis. PLoS Med. 2023;20(3):e1004188. doi:10.1371/journal.pmed.1004188
- Marchbanks PA, McDonald JA, Wilson HG, Folger SG, Mandel MG, Daling JR, et al. Oral contraceptives and the risk of breast cancer. N Engl J Med. 2002 27;346(26):2025-32. doi:10.1056/NEJMoa013202
- Wingo PA, Austin H, Marchbanks PA, Whiteman MK, Hsia J, Mandel MG, et al. Oral contraceptives and the risk of death from breast cancer. Obstet Gynecol. 2007;110(4):793-800. doi:10.1097/01.AOG.0000284446.22251.6e
- Trivers KF, Gammon MD, Abrahamson PE, Lund MJ, Flagg EW, Moorman PG, et al. Oral contraceptives and survival in breast cancer patients aged 20 to 54 years. Cancer Epidemiol Biomarkers Prev. 2007;16(9):1822-7. doi:10.1158/1055-9965.EPI-07-0053
- Nur U, El Reda D, Hashim D, Weiderpass E. A prospective investigation of oral contraceptive use and breast cancer mortality: findings from the Swedish women's lifestyle and health cohort. BMC Cancer. 2019;19(1):807. doi:10.1186/s12885-019-5985-6
- Karlsson T, Johansson T, Höglund J, Ek WE, Johansson Å. Time-Dependent Effects of Oral Contraceptive Use on Breast, Ovarian, and Endometrial Cancers. Cancer Res. 2021;81(4):1153-62. doi:10.1158/0008-5472.CAN-20-2476
- Lambe M, Hsieh C, Trichopoulos D, Ekbom A, Pavia M, Adami HO. Transient increase in the risk of breast cancer after giving birth. N Engl J Med. 1994;331(1):5-9. doi:10.1056/NEJM199407073310102
- Liu Q, Wuu J, Lambe M, Hsieh SF, Ekbom A, Hsieh CC. Transient increase in breast cancer risk after giving birth: postpartum period with the highest risk (Sweden). Cancer Causes Control. 2002;13(4):299-305. doi:10.1023/a:1015287208222
- Williams RS, Christie D, Sistrom C. Assessment of the understanding of the risks and benefits of hormone replacement therapy (HRT) in primary care physicians. Am J Obstet Gynecol. 2005;193(2):551-6. doi:10.1016/j.ajog.2005.03.061
- van Bommel MHD, IntHout J, Veldmate G, Kets CM, de Hullu JA, van Altena AM, et al. Contraceptives and cancer risks in BRCA1/2 pathogenic variant carriers: a systematic review and meta-analysis. Hum Reprod Update. 2023;29(2):197-217. doi:10.1093/humupd/dmac038
- Pearce CL, Stram DO, Ness RB, Stram DA, Roman LD, Templeman C, et al. Population distribution of lifetime risk of ovarian cancer in the United States. Cancer Epidemiol Biomarkers Prev. 2015;24(4):671-6. doi:10.1158/1055-9965.EPI-14-1128
- Kuchenbaecker KB, Hopper JL, Barnes DR, Phillips KA, Mooij TM, Roos-Blom MJ, et al. Risks of breast, ovarian, and contralateral breast cancer for BRCA1 and BRCA2 mutation carriers. JAMA. 2017;317(23):2402-16. doi:10.1001/jama.2017.7112
- Cibula D, Zikan M, Dusek L, Majek O. Oral contraceptives and risk of ovarian and breast cancers in BRCA mutation carriers: a meta-analysis. Expert Rev Anticancer Ther. 2011;11(8):1197-207. doi:10.1586/era.11.38
- Moorman PG, Havrilesky LJ, Gierisch JM, Coeytaux RR, Lowery WJ, Peragallo et al. Oral contraceptives and risk of ovarian cancer and breast cancer among high-risk women: a systematic review and meta-analysis. J Clin Oncol. 2013;31(33):4188-98. doi:10.1200/JCO.2013.48.9021
- Schrijver LH, Antoniou AC, Olsson H, Mooij TM, Roos-Blom MJ, Azarang L, et. Oral contraceptive use and ovarian cancer risk for BRCA1/2 mutation carriers: an international cohort study. Am J Obstet Gynecol. 2021;225(1):51.e1-51.e17. doi:10.1016/j.ajog.2021.01.014
- Schrijver LH, Mooij TM, Pijpe A, Sonke GS, Mourits MJE, Andrieu N, et al. Oral contraceptive use in BRCA1 and BRCA2 mutation carriers: Absolute cancer risks and benefits. J Natl Cancer Inst. 2022;114(4):540-52. doi:10.1093/jnci/djac004
- Westhoff CL, Pike MC. Hormonal contraception and breast cancer. Contraception. 2018;98(3):171-3. doi:10.1016/j.contraception.2018.05.002
- Pike MC, Spicer DV. Hormonal contraception and chemoprevention of female cancers. Endocr Relat Cancer. 2000;7(2):73-83. doi:10.1677/erc.0.0070073
- Hunter DJ. Oral contraceptives and the Small Increased Risk of Breast Cancer. N Engl J Med. 2017 Dec 7;377(23):2276-2277. doi:10.1056/NEJMe1709636
- Gérard C, Arnal JF, Jost M, Douxfils J, Lenfant F, Fontaine C, et al. Profile of estetrol, a promising native estrogen for oral contraception and the relief of climacteric symptoms of menopause. Expert Rev Clin Pharmacol. 2022;15(2):121-37. doi:10.1080/17512433.2022.2054413
- Visser M, Kloosterboer HJ, Bennink HJ. Estetrol prevents and suppresses mammary tumors induced by DMBA in a rat model. Horm Mol Biol Clin Investig. 2012;9(1):95-103. doi:10.1515/hmbci-2012-0015
- Singer CF, Bennink HJ, Natter C, Steurer S, Rudas M, Moinfar F, et al. Antiestrogenic effects of the fetal estrogen estetrol in women with estrogen-receptor positive early breast cancer. Carcinogenesis. 2014;35(11):2447-51. doi:10.1093/carcin/bgu144