How to manage endometriosis-associated pain
Pain mechanisms are multifactorial, complex, and still poorly understood.
How to manage endometriosis-associated pain | Image Credit: © Alena
- © Alena - stock.adobe.com

Endometriosis-associated pain (EAP) and the underlying mechanisms necessary for its initiation and persistence are complex and difficult to manage. This is compounded frequently by diagnostic delay. The management of EAP is multifaceted and may require a combination of hormonal and nonhormonal medications and possible surgical intervention. The purpose of this article is to review the pathophysiology of EAP, identify comorbid pain syndromes that exacerbate EAP symptoms, and propose strategies for medical management.
Pathophysiology of EAP
Endometriosis pain mechanisms are multifactorial, complex, and still poorly understood. Endometriosis implants along the pelvic viscera activate inflammatory processes and upregulate the transmission of pain signals within the central nervous system (CNS). Visceral structures (reproductive organs, bladder, bowel) share neural pathways with somatic structures (skin, muscles, fascia, bones).
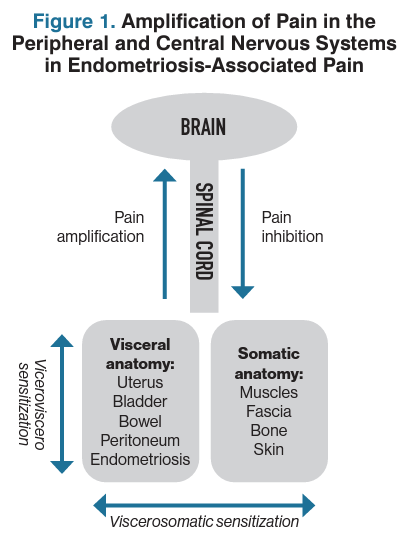
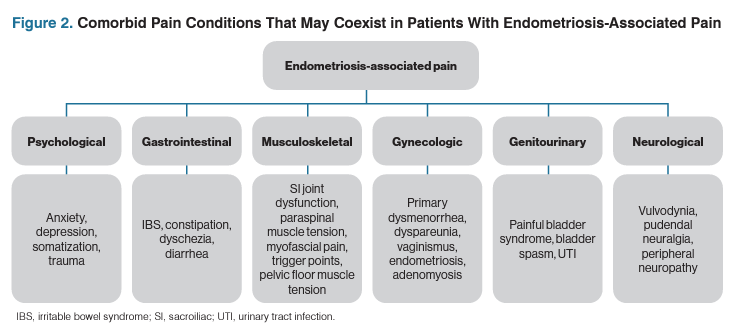
This neuropathic convergence can lead to increased sensitization of neighboring organs, increased nociceptive input to the brain and spinal cord, and an augmented CNS response to pain, therefore decreasing pain thresholds (Figure 1).1 This partially explains the multifaceted clinical symptoms present in patients with endometriosis and may explain the far-reaching symptom impact of chronic overlapping pain disorders often seen with endometriosis (Figure 2).
EAP assessment
Pain history
A detailed patient history and thorough examination can better support clinicians in the identification of comorbid pain conditions and the initiation of timely and efficacious treatment. Allotting sufficient time by scheduling a longer clinic visit or frequent follow-up visits can facilitate the multifactorial approach needed for patients with complex EAP that includes necessary education and the development of a patient-centered treatment plan.
Routine information about the onset and chronicity of pain, the relationship to menses (intramenstrual and intermenstrual), variations in severity, exacerbating/alleviating factors, character and quality, and location is relevant. Confirming prior treatment attempts and surgical interventions aimed at addressing pain may also be useful in evaluating etiologies of pain. Obtaining a social history to elucidate family dynamics, relationship status, and employment status may offer insight into the psychosocial factors that affect a patient’s pain experience. A sexual history assessing for dyspareunia (specifically with insertion, deep penetration, or orgasm), onset of occurrence, and the impact of positional changes is pertinent.
The aforementioned information, regardless of the provider’s comfort with managing every symptom, may guide individual decisions for care and the process of identifying other essential players in the multidisciplinary approach to the patient’s pain. One may consider using all or a portion of the International Pelvic Pain Society’s assessment form, which can be distributed to patients with complex pain in advance of their scheduled appointment.2
Focused physical examination
The physical examination should be performed systematically, with a trauma-informed approach (Table 1).3 The routine pelvic examination is inherently invasive and may elicit pelvic pain symptoms that trigger an unintended trauma response including agitation, dissociation, outbursts of anger, flashbacks, unconscious bodily responses, anxiety, and fear.

It is necessary for providers to create a safe and calm environment, confirm preferred pronouns and names, explain the process and purpose of each examination, obtain verbal consent prior to and throughout the exam, and inform the patient that the examination can and should be paused or terminated for any reason at their request. This effort promotes trust between the patient and provider and demonstrates respectful care. During the physical examination, the abdominal and reproductive organs as well as musculoskeletal and neurological systems should be evaluated as potential etiologies for EAP (Table 2).

Targets of pain management
Clarifying pain and fertility goals allows the transition into a modifiable care plan. Goal-setting is a necessary early-stage element in pain management, allowing for objective targets beyond a visual pain scale, and clear objectives are associated with greater satisfaction for both the physician and patient. Potential goals include functionality, coping with symptoms, minimizing dyspareunia, increasing length of pain-free or reduced-pain intervals, and pain management planning when stopping hormonal medications (ie, for desired conception). With the overarching goal toimprove quality of life, this unique, long-term therapeutic relationship with patient and provider may extend through a patient’s entire reproductive window. Regardless of the goals, a multidisciplinary, multimodal approach offers the highest likelihood of treatment success.
Nonpharmacologic interventions
Cognitive behavior therapy
Like many chronic pain conditions, psychological factors and psychiatric symptoms (eg, depression, anxiety, increased stress, and poor quality of life) frequently coexist in patients with endometriosis and may affect endometriosis-related pain perception.4 The health benefits of psychological and mind-body interventions, including mindfulness, can be implemented in patients experiencing EAP to improve pain outcomes. Cognitive behavior therapy is rooted in the belief that psychological problems are based in patterns of learned dysfunctional thinking and behaviors to a response, such as endometriosis.5
Nutrition
Oxidative stress, immunologic dysfunction, and chronic inflammation are hallmarks of endometriosis. Despite the frequent comorbid associations of gastrointestinal (GI) symptoms and food intolerances in patients with endometriosis, limited data show that dietary changes result in significant symptom relief.6,7 There is low-level evidence for pain reduction with the addition of omega-3 fatty acids due to the anti-inflammatory effects of prostaglandins PGE3 and PGE3α. Although low consumption of vitamins C, D, and E has been associated with the development of endometriosis, dietary supplementation does not appear to have a significant effect on symptoms.8 Limiting intake of low fermentable oligosaccharides, disaccharides, monosaccharides, and polyols—carbohydrates found in a wide range of foods that are challenging to digest (eg, garlic, onions, legumes, wheat, berries)—reduces symptoms in patients with endometriosis and irritable bowel syndrome. Similarly, a gluten-free diet may be associated with a reduction of symptoms for some individuals.9
Physical activity and exercise
Physical activity and exercise have long been touted as globally beneficial for pain and overall heath, with a dose-response relationship acknowledged between regular, high-intensity exercise and the effect on inflammation.10 Specific to patients with endometriosis, exercise has been associated with stress reduction and improvement in pain scores,11 although most studies lack rigorous analytic design and metrics, limiting the ability to draw strong conclusions from the existing data.12
Physical therapy
A multimodal approach that includes pelvic floor physical therapy has been proposed to improve endometriosis symptoms, although an optimal approach has not been described.13 A recent systematic review and meta-analysis describes the advantages of physical therapy in improving physical function and reducing pain intensity.14 In addition to promoting physical activity and exercise, less tangible components of physical therapy, including the incorporation of relaxation, deep breathing, and meditation techniques, are hypothesized as contributors to the physical and psychological benefits. Typically, pelvic floor physical therapy consists of weekly treatments for 8 to 12 weeks. Once stable, patients may be discharged with a home exercise program to maintain progress.
Sleep
Sleep is considered one of the few potential modifiable targets across all pain disorders. Healthy, uninterrupted, and restful sleep is required for overall brain health and cognition. Sleep dysfunction is associated with increased pain sensitivity; however, it is unknown whether this is a causal relationship. Patients with EAP are more likely to report poor sleep quality compared with control patients,15 but interventions specifically looking at sleep interventions and pain outcomes in EAP have not been reported. Sleep hygiene should be reviewed with all patients who report sleep disruptions for overall health and mood.
Acupuncture
Acupuncture is an effective and safe method of managing EAP, reducing dysmenorrhea and improving quality of life. Most studies have reported a significant reduction in pain and CA125 compared with control groups (sham or hormonal management) during treatment.16 A recent, randomized, placebo-controlled trial of acupuncture compared with sham noted a reduction in pain and improved quality-of-life metrics, but efficacy was only noted during active acupuncture treatment and did not extend once treatment was discontinued.17
Pharmacologic interventions
Analgesics
Nonsteroidal anti-inflammatory drugs (NSAIDs) are a mainstay of dysmenorrhea management, though limited data exist for management of EAP.18 They act through the nonselective inhibition of the cyclooxygenase (COX) enzyme, preventing the production of prostaglandins and thromboxane. This inflammatory blockade can be effective in the peripheral pain response. NSAIDs decrease the mucosal levels of prostaglandins (primarily driven by inhibition of COX-1), and this correlates with gastric toxicities. Because COX-2 is not constitutively expressed in the GI tract, COX-2 selective inhibitors are perceived as safer for the GI system than conventional NSAIDs.19 Additionally, COX-2 is mainly expressed in the endometrial glandular epithelium in healthy patients but is also expressed in ectopic endometriosis and has been shown to improve EAP compared with placebo in results from a small study.20 In limited quantities, NSAIDs carry minimal adverse effects (high-volume use is associated with GI toxicities, cardiovascular risks, renal injuries, hepatotoxicity, and hypertension), are readily available, and are a reasonable first-line treatment. Acetaminophen, known as the great drug potentiator, increases the effectiveness of other analgesic medications through the inhibition of central prostaglandin synthesis. Few study results have identified it as an effective pain reliever alone, so it is best when paired with other analgesics. There is limited evidence for the use of opioids for EAP; they should only be considered in refractory cases.
Hormonal medications
For patients with pain and endometriosis, suppression of menstrual cycles may be helpful with pain control (Table 3). Low-estrogenic combined oral contraceptives (COCs) are routinely used as first-line hormonal therapy in the pain management of endometriosis.18 Continuous use of monophasic pills rather than cyclic administration appears to be more effective in reducing the recurrence of dysmenorrhea but not noncyclic pelvic pain or dyspareunia. Limited clinical evidence supports a possible adverse effect of the long-term use of COCs for endometriosis pain and progression. In contrast, data from a randomized, controlled trial support the use of oral progestin-only therapy for pelvic pain associated with endometriosis and lesion suppression.21 With increasing evidence supporting progestins for pain relief, there is an argument for progestins, both oral or systemic, as first-line management of endometriosis before COCs.
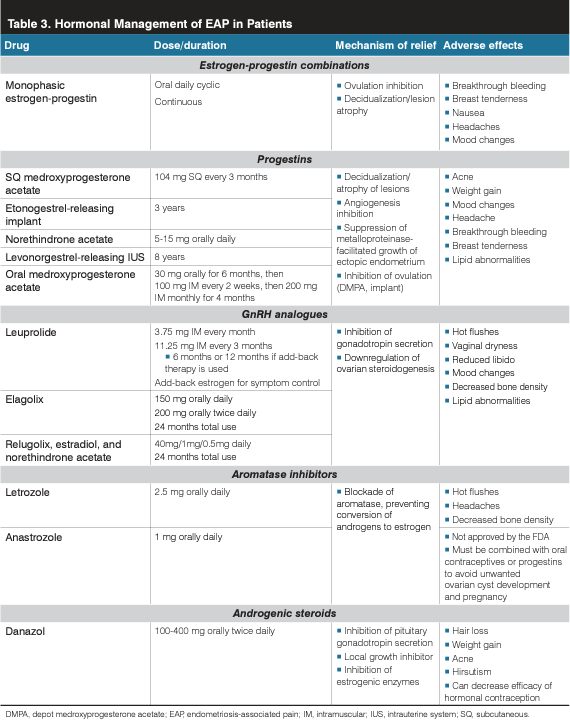
Emerging data also raise considerations for a GnRH antagonist as a reasonable second-line therapy for EAP. For patients who have had definitive surgical intervention with hysterectomy (with or without oophorectomy) and concern for persistent endometriosis lesions, aromatase inhibitors (AIs) could be considered. In premenopausal patients, AIs can be combined with progestin or COCs to reduce follicle-stimulating hormone release.22
Antidepressants
Antidepressants have been used as first-line therapy for neuropathic pain, including tricyclic antidepressants (TCAs) and serotonin-norepinephrine reuptake inhibitors (particularly duloxetine). These act by inhibiting the reuptake of serotonin and noradrenaline, thereby increasing inhibitory pain control. TCAs also act on sodium channels, beta2 adrenergic receptors, and opioid receptors. EAP, although not considered a neuropathic pain disorder, may benefit from management of the neuropathic component and comorbid psychiatric disturbances. Despite limited data in the use of antidepressants for EAP alone, patients with symptoms of endometriosis should be screened for psychosocial and psychiatric disorders. When appropriate, antidepressants may be considered for adjunct pain and mood therapy.23
Anticonvulsants
Gabapentinoids such as gabapentin and pregabalin bind to calcium channels on primary afferent nociceptors, resulting in the release of fewer neurotransmitters. Although both drugs have been broadly studied in peripheral pain syndromes, gabapentinoids have not been significantly effective in nonendometriosis chronic pelvic pain,24 though no studies have been performed exclusively for EAP. Interestingly, in endometriosis mouse models, calcium channel subunits involved in pain transmission have been identified as potential targets for reducing pain sensitivity.25 Despite this finding, gabapentinoids are frequently used and have a good safety profile with few drug interactions. In patients with multiple comorbid pain disorders, they may be considered to reduce other types of body pain.
Skeletal muscle relaxants
Skeletal muscle relaxants (SMRs) are frequently prescribed for musculoskeletal conditions, including low back and neck pain, fibromyalgia, headaches, and myofascial pain syndrome. Although popular, SMRs are not considered first-line therapy for myofascial pain and are not recommended for use beyond the management of acute muscle spasm. Myofascial pelvic pain and EAP often coexist. First-line therapy for myofascial pelvic pain remains pelvic floor physical therapy,26 although antispasmodics may be considered for musculoskeletal disorders. Antispasticity agents have activity in upper motor syndromes (eg, multiple sclerosis, cerebral palsy, traumatic brain injury, spinal cord injury, and poststroke syndrome), whereas the antispasmodics are designed for use for peripheral muscle pain and spasm. Antispastic medicines (eg, baclofen) should not be prescribed for musculoskeletal conditions because there is minimal evidence to support their use. Cyclobenzaprine is the most heavily studied SMR and has been shown to be effective for various musculoskeletal conditions, including the overlapping pain condition fibromyalgia. There are no available data specifically evaluating SMRs in EAP. If needed, the selection should be based on individual factors including potential adverse effects, desired benefits, duration of expected use, and symptom severity.
Cannabinoids
Cannabis-based products have been proposed as a novel therapeutic option for EAP. Cannabis contains delta-9-tetrahydrocannabinol and cannabidiol, which are cannabinoids that bind to endocannabinoid system receptors. Despite their popularity, there are limited high-quality data on pain outcomes in all pain conditions, including gynecologic disorders and EAP.27 In fact, emerging data suggest dysfunctional pain processing occurs in patients who use cannabis-based products, including higher levels of acute postoperative pain.
Referrals
The best approach to chronic pelvic pain is often a multidisciplinary approach. This may be necessary to achieve care metrics across the symptom spectrum in quality-of-life care. Similarly, EAP may require expanded care teams to best address chronic pain, including subspecialists in complex benign gynecology/minimally invasive gynecologic surgery, urology, functional GI medicine, physical therapy, acupuncture, pain psychology, and pain medicine.
Conclusions
The multifaceted nature of EAP explains, in part, why hormonal suppression and conservative or definitive surgical interventions may only offer a short interval of relief or incomplete symptom resolution. The education of both providers and patients regarding complex pain mechanisms and coexisting comorbid disorders highlights the importance of an equally broad approach to care.
References:
1. Impact of chronic overlapping pain conditions on public health and the urgent need for safe and effective treatment: 2015 analysis and policy recommendations. Chronic Pain Research Alliance. May 2015. Accessed March 9, 2023. http://www.chronicpainresearch.org/public/CPRA_WhitePaper_2015-FINAL-Digital.pdf
2. Documents and forms. International Pelvic Pain Society. Accessed February 2, 2023.https://www.pelvicpain.org/resources/documents-and-forms
3. Gorfinkel I, Perlow E, Macdonald S. The trauma-informed genital and gynecologic examination. CMAJ. 2021;193(28):E1090. doi:10.1503/cmaj.210331
4. Casalechi M, Vieira-Lopes M, Quessada MP, Arão TC, Reis FM. Endometriosis and related pelvic pain: association with stress, anxiety and depressive symptoms. Minerva Obstet Gynecol. 2021;73(3):283-289. doi:10.23736/S2724-606X.21.04704-3
5. Donatti L, Malvezzi H, de Azevedo BC, Baracat EC, Podgaec S. Cognitive behavioral therapy in endometriosis, psychological based intervention: a systematic review. Rev Bras Ginecol Obstet. 2022;44(3):295-303. doi:10.1055/s-0042-1742406
6. Chiaffarino F, Cipriani S, Ricci E, et al. Endometriosis and irritable bowel syndrome: a systematic review and meta-analysis. Arch Gynecol Obstet. 2021;303(1):17-25. doi:10.1007/s00404-020-05797-8
7. Schink M, Konturek PC, Herbert SL, et al. Different nutrient intake and prevalence of gastrointestinal comorbidities in women with endometriosis. J Physiol Pharmacol. 2019;70(2):10.26402/jpp.2019.2.09. doi:10.26402/jpp.2019.2.09
8. Darling AM, Chavarro JE, Malspeis S, Harris HR, Missmer SA.A prospective cohort study of vitamins B, C, E, and multivitamin intake and endometriosis. J Endometr. 2013;5(1):17-26. doi:10.5301/je.5000151
9. Marziali M, Venza M, Lazzaro S, Lazzaro A, Micossi C, Stolfi VM. Gluten-free diet: a new strategy for management of painful endometriosis related symptoms? Minerva Chir. 2012;67(6):499-504.
10. Nimmo MA, Leggate M, Viana JL, King JA. The effect of physical activity on mediators of inflammation. Diabetes Obes Metab. 2013;15(suppl 3):51-60. doi:10.1111/dom.12156
11. Goncalves AV, Barros NF, Bahamondes L. The practice of hatha yoga for the treatment of pain associated with endometriosis. J Altern Complement Med. 2017;23(1):45-52. doi:10.1089/acm.2015.0343
12. Tennfjord MK, Gabrielsen R, Tellum T. Effect of physical activity and exercise on endometriosis-associated symptoms: a systematic review. BMC Womens Health. 2021;21(1):355. doi:10.1186/s12905-021-01500-4
13. Endometriosis: diagnosis and management. National Institute for Health and Care Excellence. September 6, 2017. Accessed February 6, 2023. https://www.nice.org.uk/guidance/ng73
14. Abril-Coello R, Correyero-León M, Ceballos-Laita L, Jiménez-Barrio S. Benefits of physical therapy in improving quality of life and pain associated with endometriosis: a systematic review and meta-analysis. Int J Gynaecol Obstet. Published online December 26, 2022. doi:10.1002/ijgo.14645
15. van Barneveld E, Manders J, van Osch FHM, et al. Depression, anxiety, and correlating factors in endometriosis: a systematic review and meta-analysis. J Womens Health (Larchmt). 2022;31(2):219-230. doi:10.1089/jwh.2021.0021
16. Xu Y, Zhao W, Li T, Zhao Y, Bu H, Song S. Effects of acupuncture for the treatment of endometriosis-related pain: a systematic review and meta-analysis. PLoS One. 2017;12(10):e0186616. doi:10.1371/journal.pone.0186616
17. Li PS, Peng XM, Niu XX, et al. Efficacy of acupuncture for endometriosis-associated pain: a multicenter randomized single-blind placebo-controlled trial. Fertil Steril. 2023;S0015-0282(23)00072-9. doi:10.1016/j.fertnstert.2023.01.034
18. Brown J, Farquhar C. Endometriosis: an overview of Cochrane Reviews. Cochrane Database Syst Rev. 2014;2014(3):CD009590. doi:10.1002/14651858.CD009590.pub2
19. Rostom A, Muir K, Dubé CJ, et al. Gastrointestinal safety of cyclooxygenase-2 inhibitors: a Cochrane Collaboration systematic review. Clin Gastroenterol Hepatol. 2007;5(7):818-828. doi:10.1016/j.cgh.2007.03.011
20. Cobellis L, Razzi S, De Simone S, et al. The treatment with a COX-2 specific inhibitor is effective in the management of pain related to endometriosis. Eur J Obstet Gynecol Reprod Biol. 2004;116(1):100-102. doi:10.1016/j.ejogrb.2004.02.007
21. Casper RF. Progestin-only pills may be a better first-line treatment for endometriosis than combined estrogen-progestin contraceptive pills. Fertil Steril. 2017;107(3):533-536. doi:10.1016/j.fertnstert.2017.01.003
22. Practice bulletin no. 114: management of endometriosis. Obstet Gynecol. 2010;116(1):223-236. doi:10.1097/AOG.0b013e3181e8b073
23. Pope CJ, Sharma V, Sharma S, Mazmanian D. A systematic review of the association between psychiatric disturbances and endometriosis. J Obstet Gynaecol Can. 2015;37(11):1006-1015. doi:10.1016/s1701-2163(16)30050-0
24. Horne AW, Vincent K, Hewitt CA, et al. Gabapentin for chronic pelvic pain in women (GaPP2): a multicentre, randomised, double-blind, placebo-controlled trial. Lancet. 2020;396(10255):909-917. doi:10.1016/S0140-6736(20)31693-7
25. Bellessort B, Bachelot A, Grouthier V, et al. Comparative analysis of molecular signatures suggests the use of gabapentin for the management of endometriosis-associated pain. J Pain Res. 2018;11:715-725. doi:10.2147/JPR.S163611
26. van Reijn-Baggen DA, Han-Geurts IJM, Voorham-van der Zalm PJ, Pelger RCM, Hagenaars-van Miert CHAC, Laan ETM. Pelvic floor physical therapy for pelvic floor hypertonicity: a systematic review of treatment efficacy. Sex Med Rev. 2022;10(2):209-230. doi:10.1016/j.sxmr.2021.03.002
27. Fisher E, Moore RA, Fogarty AE, et al. Cannabinoids, cannabis, and cannabis-based medicine for pain management: a systematic review of randomised controlled trials. Pain. 2021;162(suppl 1):S45-S66. doi:10.1097/j.pain.0000000000001929

Newsletter
Get the latest clinical updates, case studies, and expert commentary in obstetric and gynecologic care. Sign up now to stay informed.
