Modernizing Treatment for Heavy Menstrual Bleeding: The Smart Evolution Into Cryoablation
Sponsored By Channel Medsystems, Inc.
Authored by Shannon Smith, MD, FACOG and Gerard Reilly, MD, FACOG
With 1 in 3 women in the US seeking treatment for heavy menstrual bleeding (HMB),1 the result is millions of patients who require tailored guidance and treatment from their physicians. Any modern gynecologic practice must identify an array of trusted HMB treatments and constantly seek meaningful advancements. At the center of it all is the need to prioritize patient needs, satisfaction, and safety.
Patient needs collide with current continuum of care
Identifying the best treatment plan begins with a patient-centered approach and shared decision-making, which starts with an honest, well-informed discussion to educate patients about different treatment options, benefits, and risks, while aligning their goals and expected outcomes to scientific evidence. Our commitment must be to seek the best fit for the patient — both for today and in the future.
First-line treatment includes conservative medical interventions with both hormonal and nonhormonal pharmacotherapy. In some instances, women are unable to use, choose not to use, or fail first-line medical therapy. Hysterectomy is one of the predominant surgical approaches to treating HMB, and, while definitive, it may carry significant complications and risks along with additional cost and resources (or resource utilization).
Global endometrial ablation (GEA) fills an important gap between medical therapy and hysterectomy for treating HMB. Compared to hysterectomy, GEA provides a minimally invasive option, with minimal risks and recovery. GEA can use heat-based or cryotherapy technologies.
Cryoablation drives the future of HMB treatment
Cryoablation presents a meaningful shift in the treatment of HMB. The evolution of cryotherapy for GEA came from a need to address many of the drawbacks of traditional heat-based thermal ablation, including intrauterine scarring and painful procedures.
The biggest difference between heat-based ablation and cryoablation is the method utilized to destroy the cells of the endometrium. Heat-based thermal ablation uses extreme heat to induce cell damage and death. Because of the healing process associated with many heat-based modalities, they often lead to scarring of the endometrial cavity with diminished cavity access post procedure, which may delay or obscure future cancer diagnosis.2
In contrast, cryothermal energy ablates the tissue without desiccation and collagen denaturation, which minimizes intracavitary scarring, adhesions, and contracture, and can help preserve future cavity access.2 Because the cooling of tissues and nerves provides an anesthetic effect, cryoablation tends to be less painful than heat-based thermal ablation techniques like microwave or radiofrequency ablation.3 As such, cryoablation is usually performed in the outpatient setting with no general anesthesia.
Physicians are recognizing this important advancement, and we are seeing widening use of cryoablation because of the many benefits it provides the patient and physician.
The Cerene® Cryotherapy Device is FDA-approved for safe and effective cryoablation
Cerene has a different tissue-healing response that minimizes intracavitary scarring, allowing physicians to access and evaluate most uterine cavities posttreatment. Additionally, the physician enjoys the simplicity, ease of use, and efficiency of a self-contained unit that allows for treatment in the office or other settings. For patients, it provides a well-tolerated treatment that can be performed in the setting that is most comfortable and familiar to them.

Meaningful patient results
The Cerene Cryotherapy Device is backed by long-term clinical outcomes, including high patient satisfaction and improved quality of life (QOL), measured up to 3 years posttreatment.4
The CLARITY trial demonstrated the efficacy of Cerene in reducing HMB symptoms and preserving cavity access. Of the 242 treated patients, 90% reported normal, light, or no periods* and 86% saw a reduction in severe and very severe dysmenorrhea.† Only 3% of patients underwent hysterectomy due to persistent HMB.
Preserved cavity access: Benefits beyond today
As physicians, our choices in treatment options must put the health and well-being of patients at the forefront. By minimizing intracavitary scarring and avoiding procedures that restrict uterine access, your future ability to evaluate endometrial pathologies is maintained. You can positively impact a lifetime of care for your patient and help protect her future diagnostic options.
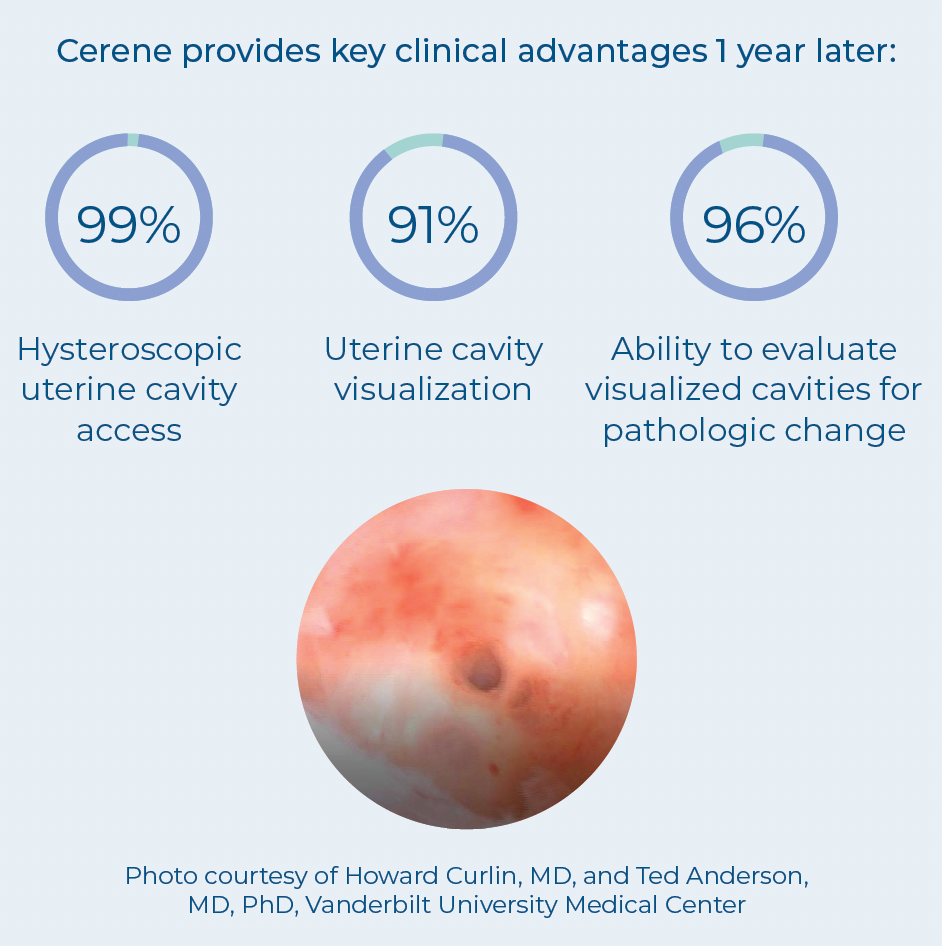
Preserving uterine access protects your ability to provide necessary evaluation should patients develop new pelvic pain or bleeding, helping to avoid a delayed or missed diagnosis of endometrial cancer. Intracavitary scarring has also been found to lead to other long-term complications, such as central or cornual hematometra, post ablation tubal sterilization syndrome (PATSS), and retrograde uterine bleeding potentially leading to the development of endometriosis.2 The American College of Obstetricians and Gynecologists (ACOG) has highlighted the importance of prioritizing ablation technologies that preserve cavity access to help us avoid diagnostic hysterectomies altogether.
QOL metrics show high patient satisfaction
Satisfaction posttreatment is a crucial measure that also deeply affects the relationship and trust between physicians and our patients. When evaluating treatment options for each patient, it’s important to understand limitations in their daily physical, home, and work activities.

Relief from dysmenorrhea
It is estimated that up to 30% of otherwise healthy women of reproductive age experience HMB with blood loss and pelvic cramping significant enough to disrupt their daily activities.5 While relief from bleeding is certainly a priority for most patients who undergo GEA, we cannot overlook dysmenorrhea that is associated with HMB. Providing relief from dysmenorrhea is a key step to getting patients back to their daily lives.

One statistic we’d like to leave you with is that 95% of patients would recommend Cerene to a friend or family member.‡ We, as physicians, are proud to recommend Cerene to our peers — because we know it works to provide relief from HMB and help preserve her diagnostic options for the future.
* Patient-reported data are 1 year after treatment with durable results at 3 years.
† Improvement reported 1 year after treatment for patients reporting severe/very severe cramping.
‡ Patient-reported data include definitely and consider recommending Cerene and are 1 year after treatment with durable results at 3 years.
To learn more, visit contact.cerene.com/contemporaryobgyn.
__________________________________
References:
1. Heavy menstrual bleeding. The American College of Obstetricians and Gynecologists Web site. https://www.acog.org/Patients/FAQs/Heavy-Menstrual-Bleeding. Updated May 2021. Accessed April 4, 2023. 2. Curlin H, Cholkeri-Singh A, Leal JGG, Anderson T. Hysteroscopic access and uterine cavity evaluation 12 months after endometrial ablation with the Cerene Cryotherapy Device [published online ahead of print November 25, 2021]. J Minim Invasive Gynecol. 2022;29(3):440-447. doi:10.1016/j.jmig.2021.11.016. 3. Erinjeri JP, Clark TWI. Cryoablation: mechanism of action and devices. J Vasc Interv Radiol. 2010;21(suppl 8):S187-S191. doi:10.1016/j.jvir.2009.12.403. 4. Curlin HL, Anderson TL. Endometrial cryoablation for the treatment of heavy menstrual bleeding: 36-month outcomes from the CLARITY study. Int J Womens Health. 2022;14:1083-1092. doi:10.2147/IJWH.S371044. 5. Kjerulff KH, Erickson BA, Langenberg PW. Chronic gynecological conditions reported by US women: findings from the National Health Interview Survey, 1984 to 1992. Am J Public Health. 1996;86(2):195- 199. doi: 10.2105/ajph.86.2.195.
Important Safety Information
Cerene® Cryotherapy Device is indicated to ablate the endometrial lining of the uterus in premenopausal women with heavy menstrual bleeding due to benign causes for whom childbearing is complete. Pregnancy following the Cerene procedure can be dangerous; therefore, contraception must be used until menopause. The Cerene procedure is not for those who have or suspect uterine cancer; have an active genital, urinary or pelvic infection; or an IUD. There are risks and considerations associated with the use of the Cerene Cryotherapy Device. Temporary side effects may include uterine cramping, vaginal infection, and lightheadedness. CAUTION: Federal (USA) law restricts this device to sale by or on the order of a physician trained in the use of the Cerene Cryotherapy Device.
©2023 Channel Medsystems, Inc. All rights reserved. Cerene and the Channel Medsystems logo are registered trademarks of Channel Medsystems, Inc. CC-23-0122 Rev A
Click to Download PDFNewsletter
Get the latest clinical updates, case studies, and expert commentary in obstetric and gynecologic care. Sign up now to stay informed.
