Surgical treatments for endometriosis
Laparoscopy remains the gold standard for diagnosis and treatment of the disease.
Endometriosis is a poorly understood condition in which endometrial-like–tissue implants outside the uterus. The disease affects 10% of reproductive-age women and 90% of patients with chronic pelvic pain and infertility.1 The mechanism of implantation and associated pain development is still incompletely described.1 Stage and pain do not correlate well because pelvic pain can be inflammatory and neuropathic in nature, with a component of central sensitization of the nervous system.1 Patients with pelvic pain exhibit enhanced anterior insula glutamatergic neurotransmission and connectivity with the prefrontal cortex, meaning that even with full excision of disease, pain may persist.2
Diagnosis
Although expert opinion suggests that early diagnosis of endometriosis is key, diagnostic challenges in combination with the requirement for a surgical procedure for diagnosis contribute to a significant delay in treatment. In 2008, US health care costs for endometriosis were estimated at $4000 annually per affected patient.1 Delays in surgical diagnosis are thought to contribute to this high cost because patients without answers seek further care and expensive testing.
Endometriosis can have serious effects on patients’ quality of life and fertility, leading to lost wages and lack of productivity. The average delay in diagnosis is approximately
7 years, preventing early treatment that can reduce chronic pain and stress on mental and emotional health.3
One must remember that chronic pelvic pain can be multifactorial, so a comprehensive evaluation for all causes of the disease must be undertaken, not only gynecologic causes, but also other organ systems, including musculoskeletal, gastrointestinal, genitourinary, and psychological. Infectious diseases as well as immunological diseases, in which the contribution to pain is more poorly understood, must also be considered. Many of these can be identified using a careful history and physical to identify a “source” organ and
through imaging.
Because of the range of etiologies for pelvic pain, a broader work-up should always be performed to rule out other causes. However, when endometriosis is suspected, further action should be taken. Although advances in imaging (eg, MRI and gel-contrast dynamic ultrasound) and the development of predictive algorithms might help with a clinical diagnosis of endometriosis, no current technology can predict all stages of the disease.4 Laparoscopy for endometriosis began in the 1970s and remains the gold standard for diagnosis and treatment of endometriosis.
Surgery for endometriosis
Given that medical therapies are generally well tolerated, most national organizations recommend a trial of medical therapy to give patients a shorter time to pain relief. Studies have shown that combined hormonal contraceptives, progestins, and gonadotropin-releasing hormone agonists are very effective at reducing pain related to endometriosis.5
However, the study authors recommend having a low threshold for surgical evaluation. For persistent pain despite a short course of medical therapy (3-6 months), surgical therapy and reevaluation of other causes of pelvic pain is indicated.
Careful patient assessment, physical examination findings, and imaging can help guide surgical management. Structural abnormalities such as adenomyosis, ovarian endometriomas, and deeply infiltrating endometriosis nodules indicate advanced disease and should expedite surgical referrals.
A comprehensive discussion regarding the limitations of surgery and continuing medical therapy and multidisciplinary treatments for pain after surgery should be had with the patient as part of preoperative counseling. Further, a discussion should take place that the finding of no endometriosis on laparoscopy and biopsy can help the physician and patient focus on identifying and treating other pain generators. The vast options for the medical suppression of endometriosis and the treatment of chronic pelvic pain are outside the scope of this manuscript but are nonetheless exceedingly important in the comprehensive management of the patient.
Excision vs ablation
Both excision and ablation of endometriosis as a means of surgical treatment remain debated. A Cochrane systematic review showed an unclear benefit with excision vs ablation, although studies were extremely heterogeneous.6 Another systematic review showed significantly greater improvement in dysmenorrhea, dyschezia, and chronic pelvic pain with excision compared with ablation procedures.7
A recent study looking at complete peritoneal excision resulted in a high level of pain relief for patients with and without endometriosis.8 Although this study is new and not comparable with patients who did not undergo surgery, the findings may shed light on the physiologic reasons why excision of the peritoneum can result in some level of pain decrease. The authors recommend performing excision surgery, with the goal being histologic diagnosis and full excision of deeply infiltrative endometriosis.8 Because lesions appear heterogeneous or can be microscopic in nature, a true diagnosis cannot be made without a biopsy. With deeply infiltrative endometriosis, the lesions extend beyond the surface of the peritoneum and require a full excision to remove.
Excision of different sites
The location and depth of lesions impact the excisional technique and the procedures performed. Endometriotic lesions are commonly found on the uterosacral ligaments, posterior cul-de-sac, and rectovaginal septum.9 The typical appearance of an endometriosis lesion is a superficial “powder-burn” implant that can be black, dark brown, or red hemorrhagic spots. Deeply infiltrating endometriosis (DIE) invades greater than 5 mm into the surrounding organ or peritoneal surface.
Figure 1. Pelvic spaces outlined after a comprehensive excision of endometriosis. Here the pararectal, paravesical, retrorectal, and rectovaginal spaces are opened to assist with removal of all disease. (Image provided by authors)
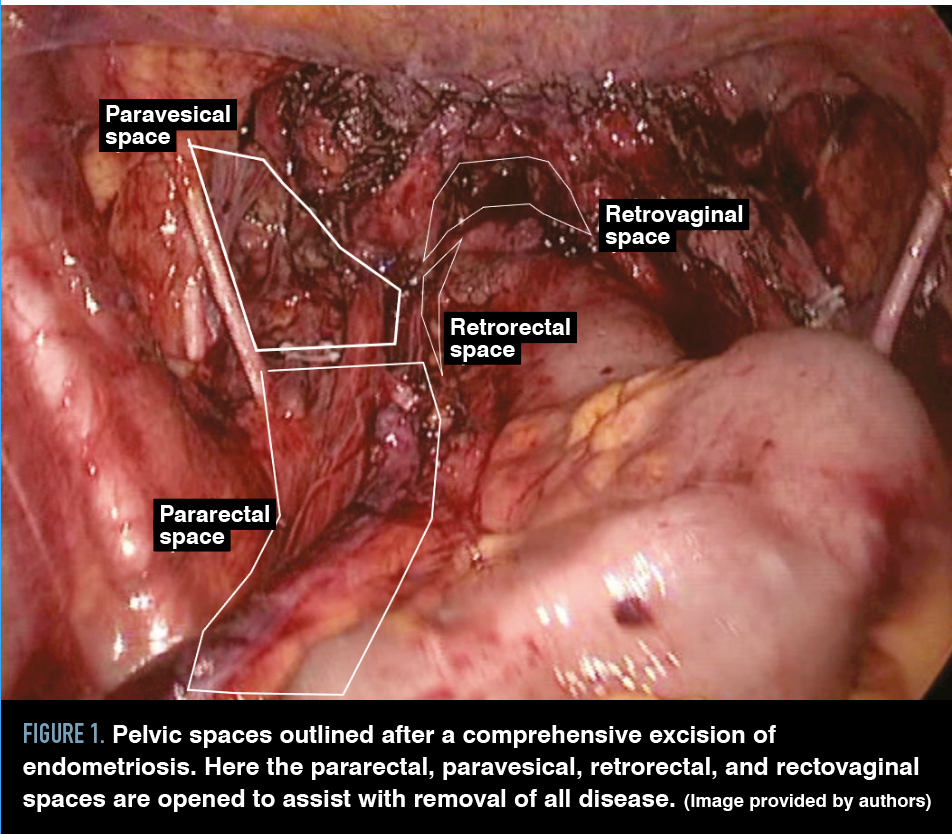
Because implants can be associated with underlying fibrosis, adhesions, and distortion of adjacent anatomy, resection must be performed with a clear understanding of anatomic structures and relevant anatomic planes, including the pararectal, paravesical, rectovaginal, retrorectal, and retropubic spaces (Figure 1 and Table).
TABLE. Pelvic Spaces and Their Borders
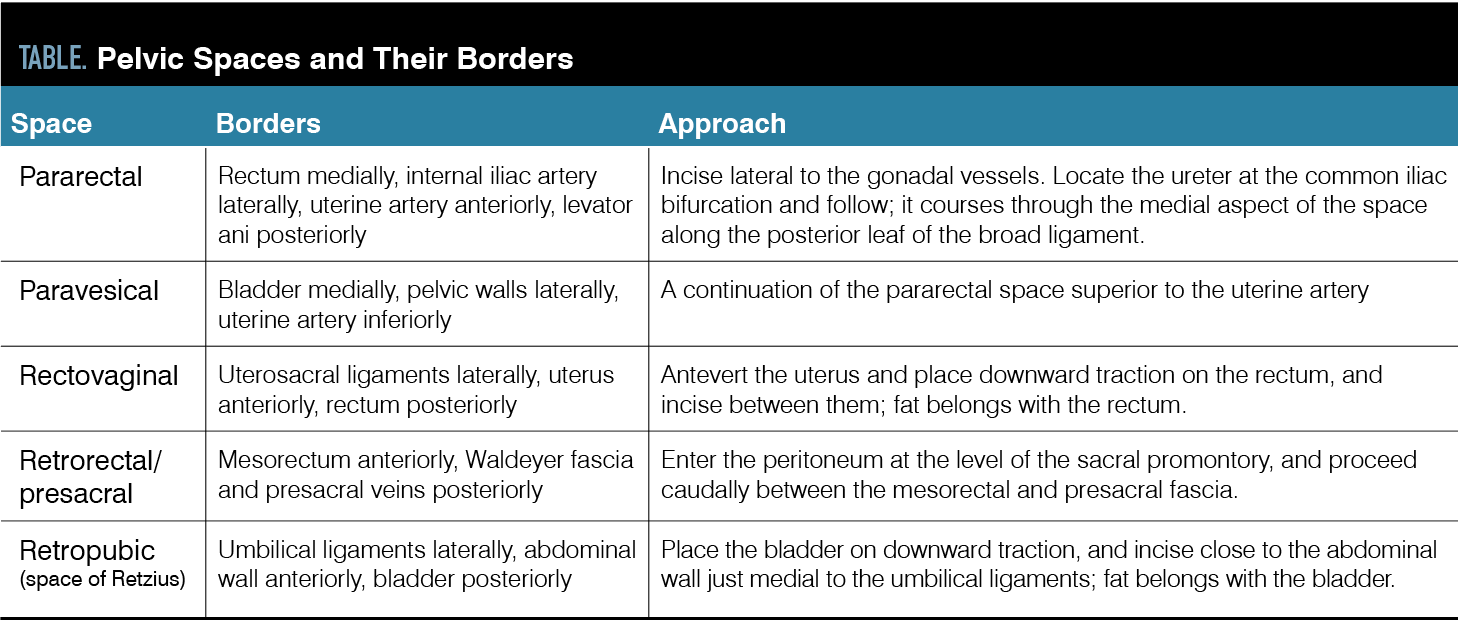
The surgeon should be comfortable with retroperitoneal dissection and both blunt and sharp dissection, with and without energy sources for lesions that lie on vital structures. Note: Because of the contractile nature of endometriosis, even early-stage disease may come close to retroperitoneal structures, including the ureter (Figure 2).
Figure 2. A) Early-stage disease in the left ovarian fossa. B) The proximity to retroperitoneal structures must always be considered. Here the left ureter dives underneath the uterine artery close to the lesion being excised. (Images provided by authors)
Complete resection results in decreased pain and recurrence compared with incomplete resection of lesions. Complete resection, however, is associated with increased complication rates, so cases involving advanced disease require meticulous technique by surgeons with substantial experience in complex endometriosis.10
Cul-de-sac lesions
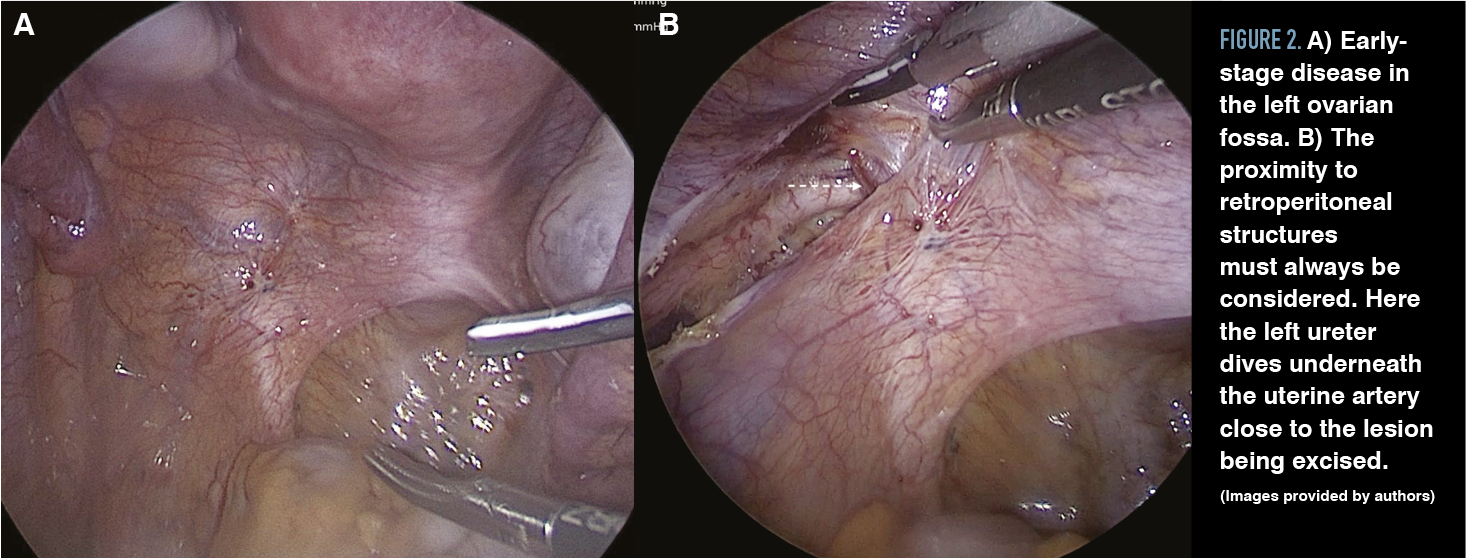
Cul-de-sac lesions are common in patients diagnosed with endometriosis, especially DIE. The cul-de-sac or the pouch of Douglas is the peritoneal cavity between the rectum and the posterior uterus. Partial obliteration of the cul-de-sac can occur when fibrotic endometriosis has fused the rectum to the posterior vagina, or complete obliteration can occur when fibrotic endometriosis obscures the plane between the cervicovaginal junction and the rectum, usually involving the uterosacral ligaments (Figures 3 and 4).
Figure 3. An MRI shows the rectum adherent to the back of the uterus with a deeply infiltrating nodule of endometriosis. (Image provided by authors)
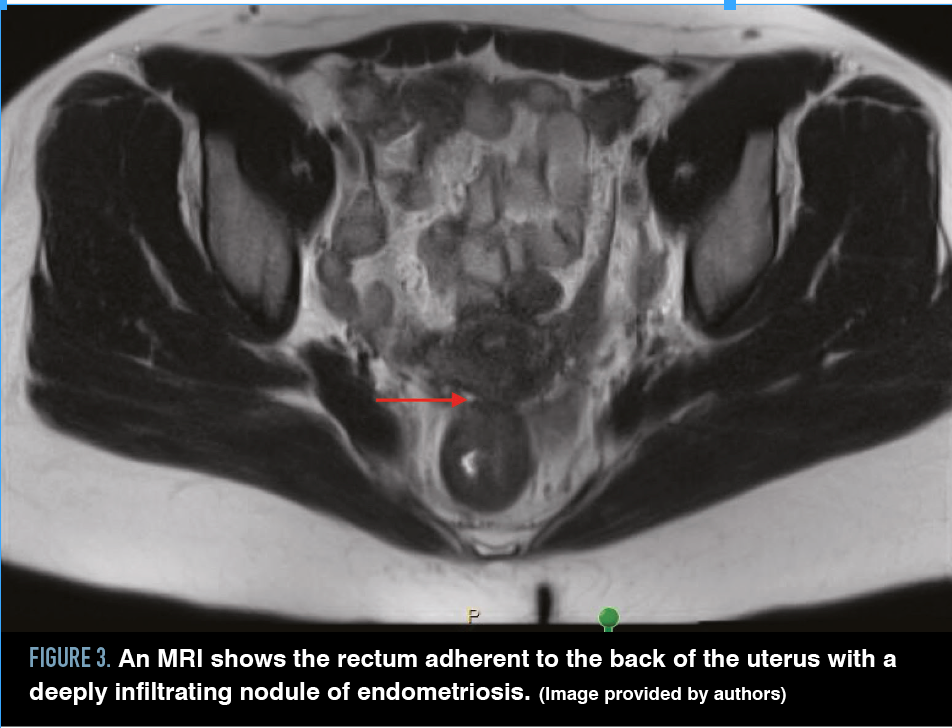
Figure 4. Surgical inspection confirms MRI findings: The rectum is stuck to the back of the uterus, and the posterior cul-de-sac is obliterated. A) Global view. B) Zoomed in. (Images provided by authors)
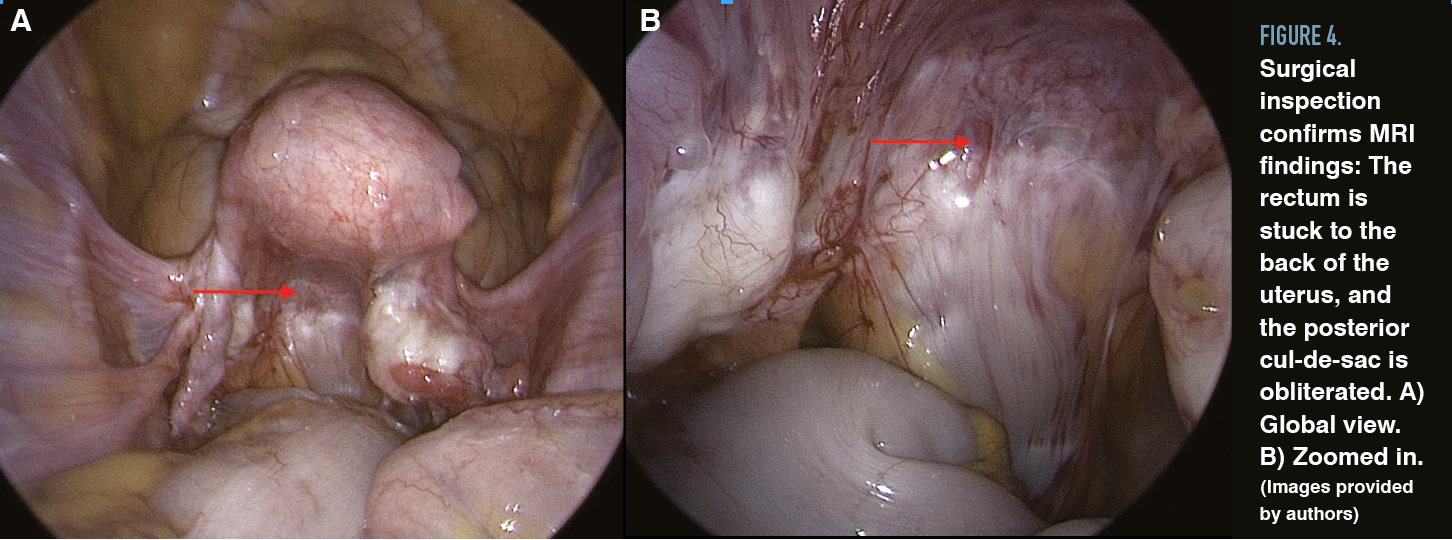
Careful dissection is needed to avoid damaging the rectum and ureters. Opening the pararectal spaces on both sides can help in dissection and restoration of normal anatomy. To open the pararectal space, an incision can be made either medially or laterally to the infundibulopelvic ligament. The ureter can be found most superficially at the bifurcation of the common internal iliac artery, and starting the dissection at the pelvic brim—where the anatomy is less distorted—can be beneficial. Early identification of anatomic structures and pelvic vasculature allows the surgeon to limit bleeding and complications.
Figure 5. A) Global anatomy has been restored. B) The rectovaginal space has been opened and the nodule of endometriosis shaved off the rectum. The rectum has been repaired with a unidirectional barbed suture. The red arrows show the sites that were previously adhered together, and the rectovaginal space is opened and identified. Note the proximity of the right ureter to the nodule that was excised. (Images provided by authors)
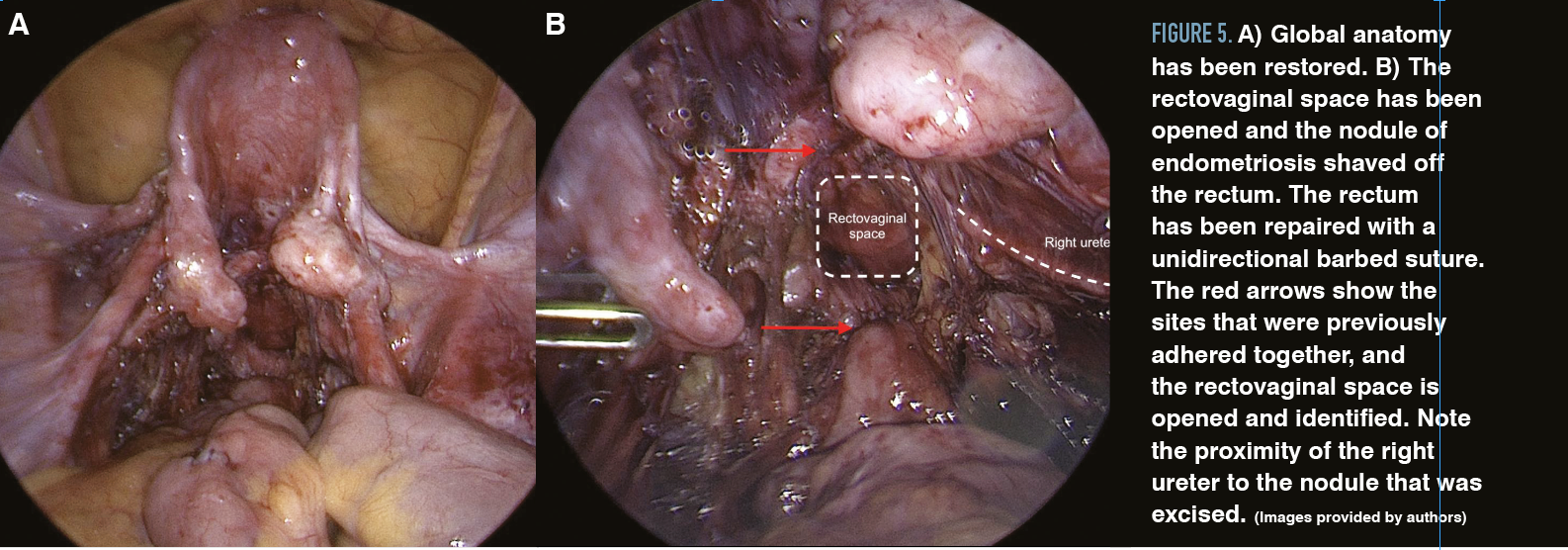
To resect lesions deep in the posterior cul-de-sac, the rectovaginal space can be entered at the uterosacral ligaments and generously developed below the fibrotic adhesions before attempting to divide them. The plane between the vagina and the rectum is avascular and can be separated with a combination of blunt and sharp dissection, up to the levator ani if necessary. Creating this plane allows the surgeon to identify the safest place to sharply divide the adhesions between the rectum and the cervix. Dilute vasopressin solution injected into the cervix and uterus prior to dissection can limit bleeding. Once the rectum has been carefully separated, the surgeon can focus on removing endometriotic implants from the vagina and cervix with electrosurgical devices (Figure 5).
Ovarian endometriomas
Ovarian endometriomas occur in 17% to 44% of patients with endometriosis.11 When treating endometriomas, the goals are to reduce pain, exclude malignancy, improve fertility, and preserve ovarian function.
Endometriomas occur when menstrual debris builds up and becomes encapsulated within ovarian tissue. The cyst does not have an epithelial lining and is considered a pseudocyst, which can make removal more difficult. Options for treatment include drainage, ablation, cystectomy, and oophorectomy. Aspiration and fenestration with ablation are surgical options; however, because of high recurrence rates with aspiration and lower effectiveness at reducing pain and improving fertility with fenestration, excision is preferred.12
For women who desire ovarian conservation, ovarian cystectomy is favored. Use of vasopressin, hemostatic agents, and suturing to achieve hemostasis results in decreased collateral damage to surrounding ovary compared with electrosurgical methods. Oophorectomy is primarily performed if patients have recurrent cysts, are postmenopausal, or there is concern for malignancy. It is important to consider that bilateral oophorectomy without hormone replacement in those younger than 50 years plays a role in increasing risks of cardiovascular disease and all-cause mortality.
Bladder endometriosis
Endometriosis can affect the urinary system, and the bladder is the most affected site. In 36% of cases, the endometriosis is localized only to the bladder. Lesions generally evolve from the serosal surface to the mucosa, and the trigone and the dome are the most affected areas.13
Prior to surgical management, it is important to rule out vesical malignancy and identify the specific location of the lesion, including distance from the ureter. Complete resection of these lesions has been associated with decreased recurrence and significant reduction of pain.13
Treatment for the gynecologist typically involves partial cystectomy with full thickness incision. For lesions near the trigone, ureteral stents should be employed. Mobilization of the retropubic space can aid in visualization of the borders of the lesion. Closure of the cystotomy depends on the size of the lesion; however, as with repair of an inadvertent bladder injury, 2-layer closure using a 3-0 delayed absorbable suture can generally be utilized.14
Rectal endometriosis
When endometriosis involves the rectosigmoid colon, the depth of invasion should be assessed. Disease severity can vary greatly, from superficial adhesions to full-thickness disease that penetrates the mucosa. When gastrointestinal involvement is suspected, MRI, gel-contrast ultrasonography, and colonoscopy with endoscopic ultrasound can help assess colonic involvement. For severe disease—including cases with multiple bowel implants, sigmoid involvement, lesions greater than 3 cm, and lesions that involve greater than 50% of the bowel circumference, or in cases with bowel stenosis—resection with reanastomosis is beneficial.14
For more superficial cases, a shaving technique or discoid resection can be performed. A shaving technique can be implemented with sharp dissection of the implant down to the level of the muscularis. The bowel can then be oversewn perpendicular to the incision to avoid constriction of the bowel lumen. Barbed suture has been shown to be safe when suturing the bowel; however, traditional delayed absorbable sutures can also be used.15 In patients with smaller nodules that involve the mucosa, discoid resection can be considered, with closure in the same fashion.
In one study comparing the shaving technique, discoid resection, and segmental resection, the shaving group had a higher rate of recurrence of dysmenorrhea and dyspareunia, with similar rates of postoperative complications.
Lesions greater than 3 cm were associated with an increased risk of requiring bowel resection.16
Integrity of the bowel after a rectovaginal dissection or bowel resection should be confirmed. In a “bubble test,” air is injected retrograde into the rectum, the sigmoid is occluded at the sacral promontory, and the pelvis is filled with sterile irrigant. Observing bubbles indicates an opening that must be addressed to prevent postoperative bowel leakage into the peritoneal cavity.
Abdominal wall endometriosis
Endometriosis of the abdominal wall is rare but expected to increase with rising cesarean section rates.17 In most studies, cesarean section was the inciting surgery; however, cases following laparotomy and port-site endometriosis have also been described, as well as cases in patients with no history of prior abdominal surgery. Some studies show concurrent pelvic endometriosis, whereas others describe cases with no associated pelvic lesions.17
Excision of the implant with complete margins is recommended to decrease risk of recurrence. Surgery may be booked with a general surgeon for closure if a large fascial defect is anticipated. Preoperative radioisotope injection to clearly identify small lesions has been described but is not well studied.18
Extrapelvic endometriosis
Extrapelvic endometriosis is a rare condition, representing 8.9% of endometriotic cases. The most common sites include the gastrointestinal tract, urinary tract, and distant organs including the lung, abdominal wall, liver, gallbladder, and pancreas. Thoracic endometriosis syndrome, although rare, is the most frequent extrapelvic type of endometriosis. Catamenial pneumothorax (recurrent pneumothorax associated with menstruation) is a possible manifestation. Video-assisted thoracoscopic surgery to remove all endometriotic lesions and pleurectomy or pleurodesis are recommended
treatment strategies.19
Further management
Although surgical management of endometriosis is considered the gold standard for diagnosis and treatment of the disease, it should not be considered curative of all chronic pelvic pain. Pelvic pain can be both neuropathic and inflammatory in nature, with central sensitization of the nervous system and prefrontal cortex remodeling leading to continued pain even after excision of all endometriosis.2
Discouragingly, 30% of patients have chronic pain that is not responsive to surgery, and 50% develop recurrent symptoms within 5 years of treatment.5 One study found high pain recurrence rates after operative laparoscopy in patients aged 19 to 29 years, despite having no identifiable lesions on
repeat surgery.20
Patients with continued pain after surgery need comprehensive multidisciplinary management for their symptoms, including continued medical suppression of menses, consultations with pelvic floor physical therapists and pain interventionalists, and referrals to other subspecialists for the other organ systems involved.
Future directions
To improve diagnosis of endometriosis and preoperative planning, imaging with an experienced sonographer can identify areas of endometriotic disease through site-specific tenderness, fixed ovaries, a negative “sliding sign,” and evidence of DIE nodules in the anterior and posterior compartments.21 Further, advances in MRI-acquisition protocols have improved diagnosis for DIE and enhancing lesions.22 Improved imaging can be diagnostic and also provide guidance for the surgeon in removing all the disease.
Conclusion
Endometriosis is a heterogeneous disease that inflicts a significant quality-of-life and cost burden on patients. Laparoscopy remains the gold standard for diagnosis and treatment. Although it is not curative for all chronic pelvic pain, laparoscopy should be a start in the approach to patients with suspicious symptoms. We recommend a comprehensive work-up for all causes of pelvic pain, and when endometriosis is suspected, preoperative planning with imaging, when applicable, and excision of all disease. When DIE is suspected, treatment should include referral to a subspecialist with experience in complex endometriosis.
wReferences
- Zondervan KT, Becker CM, Missmer SA. Endometriosis. N Engl J Med. 2020;382(13):1244-1256. doi:10.1056/NEJMra1810764
- As-Sanie S, Kim J, Schmidt-Wilcke T, et al. Functional connectivity is associated with altered brain chemistry in women with endometriosis-associated chronic pelvic pain. J Pain. 2016;17(1):1-13. doi:10.1016/j.jpain.2015.09.008
- Taylor HS, Adamson GD, Diamond MP, et al. An evidence-based approach to assessing surgical versus clinical diagnosis of symptomatic endometriosis. Int J Gynaecol Obstet. 2018;142(2):131-142. doi:10.1002/ijgo.12521
- Nnoaham KE, Hummelshoj L, Kennedy SH, Jenkinson C, Zondervan KT; World Endometriosis Research Foundation Women’s Health Symptom Survey Consortium. Developing symptom-based predictive models of endometriosis as a clinical screening tool: results from a multicenter study. Fertil Steril. 2012;98(3):692-701.e5. doi:10.1016/j.fertnstert.2012.04.022
- Practice bulletin no. 114: management of endometriosis. Obstet Gynecol. 2010;116(1):223-236. doi:10.1097/AOG.0b013e3181e8b073
- Bafort C, Beebeejaun Y, Tomassetti C, Bosteels J, Duffy JM. Laparoscopic surgery for endometriosis. Cochrane Database Syst Rev. 2020;10(10):CD011031. doi:10.1002/14651858.CD011031.pub3
- Pundir J, Omanwa K, Kovoor E, Pundir V, Lancaster G, Barton-Smith P. Laparoscopic excision versus ablation for endometriosis-associated pain: an updated systematic review and meta-analysis. J Minim Invasive Gynecol. 2017;24(5):747-756. doi:10.1016/j.jmig.2017.04.008
- Misal M, Girardo M, Wasson M. Improved pain and quality of life after complete pelvic peritonectomy. Am J Obstet Gynecol. 2022;226(3):S1273-S1274. doi:10.1016/j.ajog.2021.12.165
- Lee HJ, Park YM, Jee BC, Kim YB, Suh CS. Various anatomic locations of surgically proven endometriosis: a single-center experience. Obstet Gynecol Sci. 2015;58(1):53-58. doi:10.5468/ogs.2015.58.1.53
- Cao Q, Lu F, Feng WW, Ding JX, Hua KQ. Comparison of complete and incomplete excision of deep infiltrating endometriosis. Int J Clin Exp Med. 2015;8(11):21497-21506.
- Zanelotti A, Decherney AH. Surgery and endometriosis. Clin Obstet Gynecol. 2017;60(3):477-484. doi:10.1097/GRF.0000000000000291
- Levy B, Barbieri R. Endometriosis: management of ovarian endometriomas. UpToDate. Accessed October 12, 2022. https://www.uptodate.com/contents/endometriosis-management-of-ovarian-endometriomas
- Maccagnano C, Pellucchi F, Rocchini L, et al. Diagnosis and treatment of bladder endometriosis: state of the art. Urol Int. 2012;89(3):249-258. doi:10.1159/000339519
- Baggish MS, Karram M. Atlas of Pelvic Anatomy and Gynecologic Surgery. 5th ed. Elsevier Inc; 2020.
- Chamsy D, King C, Lee T. The use of barbed suture for bladder and bowel repair. J Minim Invasive Gynecol. 2015;22(4):648-652. doi:10.1016/j.jmig.2015.01.030
- Afors K, Centini G, Fernandes R, et al. Segmental and discoid resection are preferential to bowel shaving for medium-term symptomatic relief in patients with bowel endometriosis. J Minim Invasive Gynecol. 2016;23(7):1123-1129. doi:10.1016/j.jmig.2016.08.813
- Carsote M, Terzea DC, Valea A, Gheorghisan-Galateanu AA. Abdominal wall endometriosis (a narrative review). Int J Med Sci. 2020;17(4):536-542. doi:10.7150/ijms.38679
- Vitral GSF, Salgado HC, Rangel JMC. Use of radioguided surgery in abdominal wall endometriosis: an innovative approach. World J Nucl Med. 2018;17(3):204-206. doi:10.4103/wjnm.WJNM_47_17
- Chamié LP, Ribeiro DMFR, Tiferes DA, Macedo Neto AC, Serafini PC. Atypical sites of deeply infiltrative endometriosis: clinical characteristics and imaging findings. Radiographics. 2018;38(1):309-328. doi:10.1148/rg.2018170093
- Stratton P, Berkley KJ. Chronic pelvic pain and endometriosis: translational evidence of the relationship and implications. Hum Reprod Update. 2011;17(3):327-346. doi:10.1093/humupd/dmq050
- Noventa M, Scioscia M, Schincariol M, et al. Imaging modalities for diagnosis of deep pelvic endometriosis: comparison between trans-vaginal sonography, rectal endoscopy sonography and magnetic resonance imaging. a head-to-head meta-analysis. Diagnostics (Basel). 2019;9(4):225. doi:10.3390/diagnostics9040225
- Lorusso F, Scioscia M, Rubini D, et al. Magnetic resonance imaging for deep infiltrating endometriosis: current concepts, imaging technique and key findings. Insights Imaging. 2021;12(1):105. doi:10.1186/s13244-021-01054-x
Advancing obstetric and gynecologic surgery through robotic innovation
April 30th 2024Explore how robotic surgery revolutionizes obstetric and gynecologic procedures, offering enhanced precision, reduced complications, and improved patient outcomes, while also examining challenges and future prospects in the field.
Read More
Unlocking placenta accreta spectrum with single-cell gene targets
April 18th 2024Discover how cutting-edge single-cell RNA sequencing unveils molecular insights into placenta accreta spectrum disorders, potentially revolutionizing diagnostics and treatments for this life-threatening pregnancy complication.
Read More
Excessive gonadotropins in IVF: Effects on mosaicism and live birth
April 12th 2024A recent study revealed a correlation between high doses and prolonged duration of exogenous gonadotropin use during in vitro fertilization and increased embryonic mosaicism alongside diminished live birth rates, prompting reconsideration of dosage and duration protocols.
Read More
Maternal history linked to uterine fibroid risk in Black women
April 11th 2024Delve into the findings of a recent study revealing the heightened risk of developing uterine fibroids among Black women with a maternal history of the condition, shedding light on crucial implications for patient care and advocacy.
Read More

