
Newly published data from a phase 3 study demonstrate the safety and efficacy of fezolinetant (Astellas) for treatment of VMS due to menopause.

Newly published data from a phase 3 study demonstrate the safety and efficacy of fezolinetant (Astellas) for treatment of VMS due to menopause.

Covis, the manufacturer of the controversial preterm birth prevention drug Makena, has announced it will voluntarily withdraw the drug from the market after recommendations from the Center for Drug Evaluation and Research.
The therapy, bepirovirsen, is an antisense oligonucleotide that targets all Hepatitis B (HBV) messenger RNAs, and the company is hoping it will become a functional cure.
Individuals that did not respond had a lower percentage of naïve T cells compared to responders, while responders had a lower percentage of Th2 and CD4 central memory subsets before vaccination.
The wearable device received its original FDA go-ahead in 2019, and has since demonstrated effectiveness in preventive and acute care for migraine treatment in adolescents and adults.

The federal agency set a Prescription Drug User Fee Act (PDUFA) action date of August 2023.

Results reported at CROI showed that 2 cohorts of the ongoing, phase 3 DELIVER study that this form of PrEP modality posed no differences in pregnancy and infant outcomes.
A look at contraceptive methods and how they are poised to affect women’s health.

The FDA has granted a regular approval to dostarlimab-gxly (Jemperli; GSK) for adult patients with mismatch repair–deficient, recurrent or advanced endometrial cancer that has progressed on or after a prior platinum-containing regimen in any setting and who are not candidates for curative surgery or radiation.
The US Food and Drug Administration has accepted the Biologics License Application (BLA) for a proposed biosimilar of denosumab (Prolia and Xgeva; Amgen) for treatment of various conditions.

The rare condition can lead to life-threatening anemia in the fetus and currently lacks FDA-approved treatments.

In this interview, Michael Krychman, MD, a community-based gynecologist in Newport Beach, California, takes a look at 2022 advances within the contraceptive space, as well as looks toward the future.
Marco Taglietti, MD, president and CEO of Scynexis, sits with Contemporary OB/GYN® to discuss the recent approval of ibrexafungerp (Brexafemme) for the reduction in incidence of recurrent vulvovaginal candidiasis.
The precision medicine company announced successful completion of its Unyvero urinary tract infection (UTI) clinical trial with promising results and plans for submission to the FDA.

GSK plc has announced positive results for safety and tolerance of dostarlimab (Jemperli; GSK plc) in treating women with endometrial cancer.

Ibrexafungerp (Brexafemme; Scynexis) is now approved for the treatment of vulvovaginal candidiasis and reduction in the incidence of recurrent vulvovaginal candidiasis (RVVC).

This is the first FDA-approved ADC for platinum-resistant disease, according to ImmunoGen.

Daré Bioscience announced yesterday that it has received a grant from the Bill & Melinda Gates Foundation of $584,986 to support its efforts related to the development of a vaginal thermosetting gel formulation for the delivery of live biotherapeutics that can be reconstituted at the point of care.
The Food and Drug Administration (FDA) has approved Medicine360’s supplemental new drug application to extend the use of its levongestrel-releasing intrauterine system 52mg (LILETTA) to prevent pregnancy for up to 8 years.
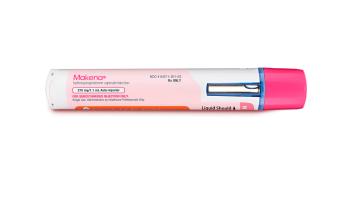
An advisory panel for the US Food and Drug Administration voted 14-1 to withdraw 17α-hydroxyprogesterone caproate (Makena; Covis Pharma) from the market.

The controversial 17α-hydroxyprogesterone caproate (17-OHPC; Makena) showed to be ineffective at preventing preterm birth, according to briefing documents.
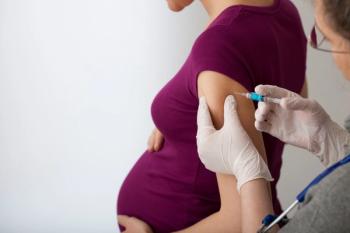
The US Food & Drug Administration approved tetanus toxoid, reduced diptheria toxoid and acellular pertussis vaccine, adsorbed [Tdap] (Boostrix, GSK) today for immunization during the third trimester of pregnancy to prevent pertussis—or whooping cough—in infants under 2 months of age.

Nkechi Azie, MD, MBA, FIDSA, vice president of clinical development and medical affairs at SCYNEXIS, discusses the potential FDA approval of ibrexafungerp (Brexafemme; SCYNEXIS) for the prevention of recurrent vulvovaginal candidiasis.
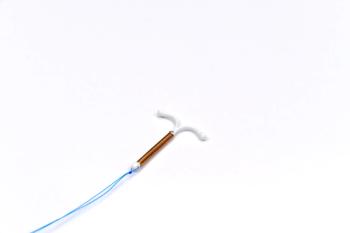
After a recent FDA approval, Mirena can be used to prevent pregnancy for up to 8 years.
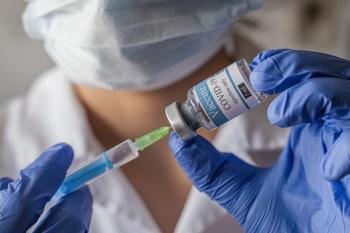
Novavax joins the growing list of COVID-19 vaccines including vaccines from Pfizer-BioNTech, Moderna, and Johnson & Johnson’s Janssen