
A study charts how the body's fat and lean mass change during the transition.

A study charts how the body's fat and lean mass change during the transition.

Disruptions in circadian stimuli resulting from their work schedule may put nurses under age 45 at higher risk for accelerated onset of menopause.

The timing of a woman's cardiovascular event may play a prominent role in her menopause onset.
Researchers in Israel looked at the effectiveness of hysteroscopy compared with TVUS in diagnosing polyps in postmenopausal women.
New results from a prospective cohort study conflict with finding from previous studies regarding MHT and cutaneous melanoma risk.

A recently published cross-sectional analysis examined how common IPV, sexual assault, and PTSD are among this population and whether these exposures are associated with a woman’s menopause experience.

Data from the SWAN study underscore how critical midlife is to CVD health.
A recent study calls into question the need for a "black-box" warning on this formulation.

Despite the prevalence of GSM, the condition continues to be under-recognized and undertreated due to a combination of patient and provider factors. Providers need to take the initiative in screening and patient education.

The first birth from a groundbreaking trial of in utero stem cell transplantation suggests that fetal therapy may be a viable new option for alpha thalassemia (ATM). Plus: Why do vasomotor symptoms bother some women long after menopause? Also: Does CPAP improve sexual QOL in women with sleep apnea?

Results of a recent extended study of prolapse surgery showed that in 60% of women, two common procedures failed within 5 years, but patients still reported a higher quality of life than before the surgery. Plus: ACOG has released a revised Committee Opinion to emphasize the idea of the “fourth trimester” in an attempt to reduce maternal morbidity and mortality numbers. Also: A recent study suggests that entering menopause later in life may be associated with a small boost in memory performance years later.


Guidelines from the National Institute for Health and Care Excellence (NICE) offer a unique perspective on diagnosis and management of menopause. Plus: A guide for hormone replacement therapy.

A study looks at the efficacy of aspirin in reducing preterm preeclampsia. Plus: The CDC reports on antidepressant use in the United States. Also, a look at whether vaginal estrogen postmenopause increases the risk of heart disease and certain types of cancer.

This article reviews how migraines in women are impacted by the fluctuating hormone levels they experience throughout their lifecycle.

A first-of-its-kind capsule combining natural 17β-estradiol and progesterone has promise for menopause-related hot flushes, according to results of a Phase 3 multi center trial.

Dr Greenberg reports his thoughts on the Sureglide device and preventing lacerations during cesarean, Überlube and helping women with vaginal dryness and dyspareunia, and TruClear ULTRA Mini tissue removal device for mechanical morcellation.

Can adopting quality measures decrease the cesarean rate? Plus: The Government Accountability Office issues a report on power morcellators and the FDA's system of evaluation. Also, does the volume of one type of heart fat indicate the risk of heart disease?

2016 was a year of change and new challenges. Find out what the top news stories of 2016 were.

A study examines the efficacy of lower-than-conventional doses of estrogen in relieving vasomotor symptoms. Plus: What are the latest ACOG recommendations on water births?

A new study looks at the impact of hormone replacement therapy on preventing Alzheimer's. Plus: Are breast density readings actually accurate?

Are women who exposed to flame retardants at greater risk of thyroid disease? Plus: A look at the potential connection between maternal smoking status and risk of the child developing schizophrenia. Also, can telephone therapy help menopausal women get better sleep?

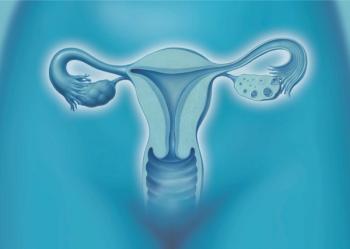
A new study looks at whether ovary-sparing hysterectomies can lead to early menopause. Plus: Does betamethasone reduce respiratory complications in preterm infants? Also, the impact of the vaginal microbiome on preterm birth.

The FDA approves the first containment system for morcellation. Plus: ASRM provides guidance for Zika virus. And, the impact of HRT timing and its impact on cardiovascular disease.

Do women suffer from more migraines as they transition into menopause? Plus: A report indicates that hospitals may not be adequately supporting breastfeeding in teenaged mothers. And, a look at prenatal exposure to bronchodilators and autism.