Vulvovaginal disease in the inpatient setting: Part 2
In part 2 of this review, more vulvovaginal diseases are examined.
EDITOR’S NOTE: This is part 2 of a 2-part series. Part 1 was published in the July issue of Contemporary OB/GYN.
Skin disease is common in hospitalized patients. This article focuses on the recognition and management of common dermatologic and systemic diseases that may present with vulvovaginal involvement. In Part I, we reviewed red flags for severe skin disease. Then we reviewed drug-induced conditions where particular care should be taken to manage vulvovaginal involvement in the inpatient setting. Here, we continue our discussion and cover vulvovaginal involvement in inflammatory and ulcerative conditions in the inpatient setting.
Inflammatory conditions
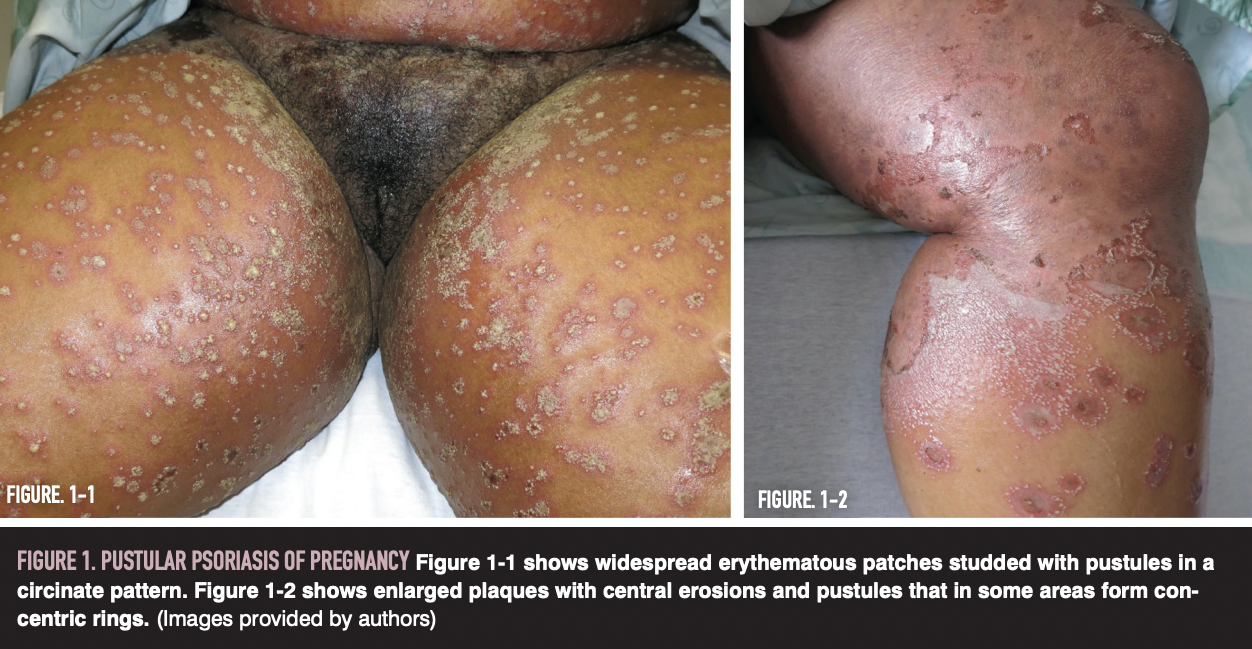
Pustular psoriasis of pregnancy
Pustular psoriasis of pregnancy (PPP) is a life-threatening condition that typically occurs in the third trimester of pregnancy, though it may present during any trimester.
Patients present with symmetric erythematous plaques with overlying sterile cutaneous pustules at plaque margins in a circinate pattern (Figure 1). Skin changes are often accompanied by systemic signs and symptoms such as fatigue, fever, elevated inflammatory markers, diarrhea, and delirium.1-3 In severe cases, disease can progress to erythroderma and be complicated by dangerous fluid and electrolyte imbalances, loss of thermoregulation in the skin, and secondary infection and sepsis.1,4
Evidence for treatment of patients with PPP is limited and depends on severity of presentation. First-line treatments include high-dose systemic corticosteroids vs low-dose cyclosporine.1 This condition is associated with placental insufficiency, so systemic signs and symptoms need close fetal monitoring because of an increased risk of fetal anomalies and fetal demise.1,5 Although pustular psoriasis of pregnancy often resolves after parturition, cases have been reported in the postpartum period, and recurrence in subsequent pregnancies is common.1,6
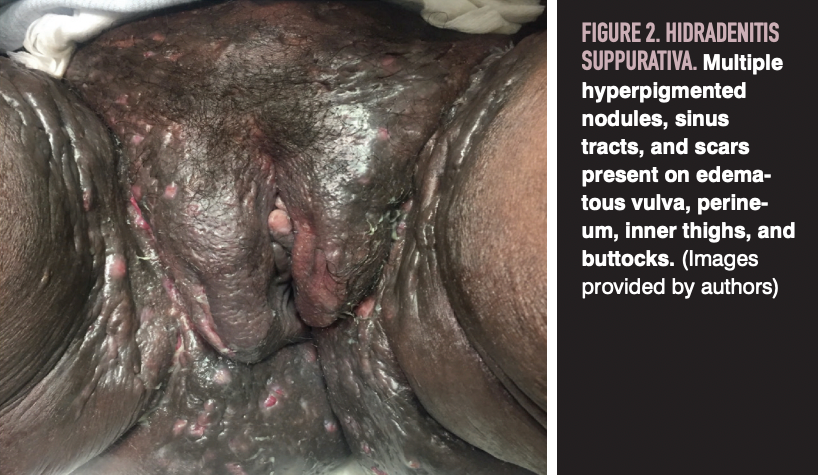
Hidradenitis suppurativa
Hidradenitis suppurativa (HS) is a chronic follicular occlusive disease that affects any skin area containing folliculopilosebaceous units. Usual symptom onset is in the second or third decade of life, and females are more likely to be affected.7-9 Factors associated with the disease include a family history of HS, obesity, and smoking.
The primary lesion of HS is the recurrent, painful inflammatory nodule, which predominantly affects intertriginous areas. Distribution is influenced by sex, and females are more likely to have involvement of the axillae, upper anterior torso including breast and inframammary regions, buttocks, and gluteal cleft.10,11
HS may also affect the vulva (Figure 2). Vulvar HS can often be mistaken for other vulvar diseases such as abscesses, folliculitis, or Bartholin cyst.12 Compared with patients with HS without vulvar involvement, patients with vulvar HS tend to present at a later onset, with lower body mass index, and higher incidence of psychiatric disease.12 Clinical manifestations include the presence of fistulas with minimal axillary involvement.12 Chronic disease may result in subcutaneous extension with suppuration, sinus tract formation, or hypertrophic scarring. In addition, an association with vulvar cancer has been demonstrated in patients with disease involving the groin.13
Treatment depends on disease stage. Acute management includes education, topical field therapy, anti-inflammatory antibiotics, intralesional triamcinolone (Kenalog), or prednisone as a bridge to biologics. Most patients with vulvar HS have been shown to respond well to medical treatment and not require surgery.12 HS may significantly impact sexual function and quality of life. In cases of anogenital involvement, impairment can be especially significant.14 Patient support is available at the HS Foundation website.
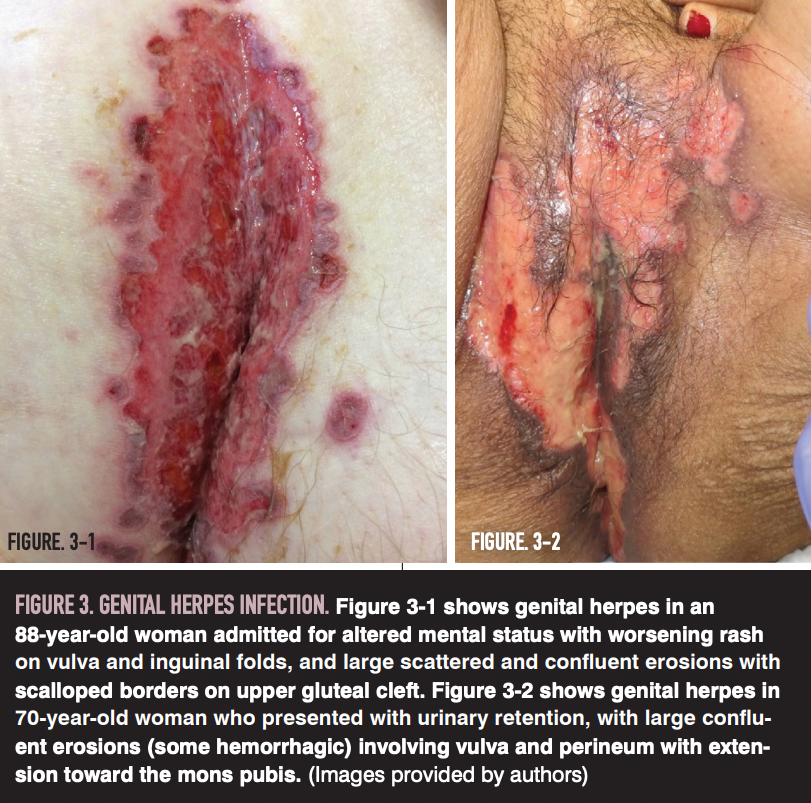
Genital herpes
Genital herpes is caused by primary and recurrent infection involving herpes simplex virus type 1 (HSV-1) or type 2 (HSV-2). Most cases of recurrent disease are caused by HSV-2, but increasingly HSV-1 is associated with genital herpes, especially in younger women.15 Genital herpes is underdiagnosed because infection is often subclinical.16
In women, genital herpes commonly involves the vulva and vagina (Figure 3). Primary infection presents with highly variable symptoms, including painful genital ulcers, systemic symptoms such as fever and headache, local pain and pruritus, tender lymphadenopathy, and dysuria.17-19 Physical exam in primary disease reveals characteristic grouped 2- to 4-mm vesicles, erosions, or ulcers, which may be angulated or linear. Other patients have milder symptoms or may be asymptomatic. Recurrent infection is characterized by less severe symptoms, including smaller vesicular or ulcerative lesions that are often unilateral.18 Of note, rate of recurrent infection is positively associated with initial infection duration and severity, but unrelated to treatment with acyclovir.20
Clinical diagnosis is neither sensitive nor specific, and suspicions should be followed with routine use of virological and type-specific serology. Viral culture and polymerase chain reaction testing are preferred methods in cases of active lesions. Most patients experiencing their first episode of genital HSV infection should be treated with an antiviral to decrease duration and severity of disease. Patients with severe HSV disease or complications, such as disseminated infection, pneumonitis, or hepatitis, should be treated with intravenous acyclovir until clinical improvement, with subsequent oral antivirals to total at least 10 days of therapy.21 Treatment of recurrent infection should be individualized; chronic suppressive therapy is generally recommended in patients with severe or frequent recurrences. In addition, all patients with genital herpes should be counseled on the natural course of the disease, including identifying any symptomatic prodrome prior to recurrence and understanding the potential for asymptomatic transmission.
Ulcerative conditions
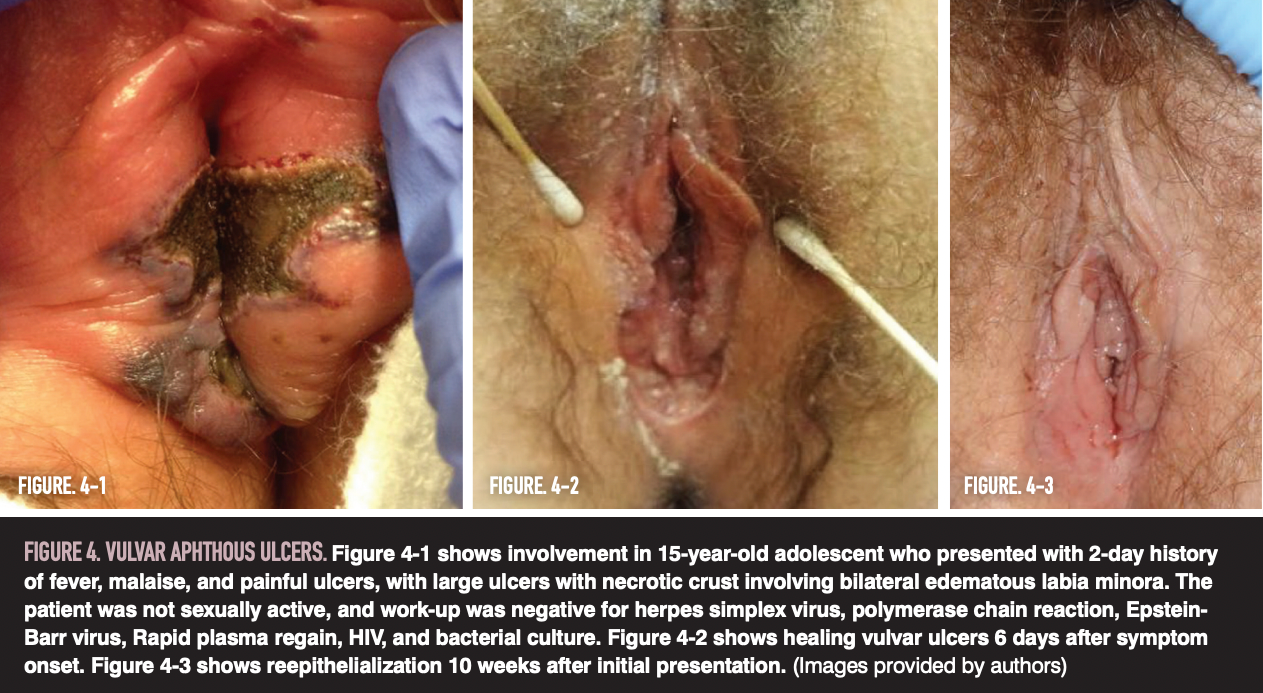
Acute vulvar aphthous ulcers
Acute vulvar aphthous ulcers (also known as Lipschütz acute vulvar ulceration) are similar to the more common oral aphthous ulcer, though typically much larger in size.22 The condition mainly affects patients 20 years or younger, who present with sudden onset of necrotic and painful ulcers. Exact disease pathogenesis is not certain but may be related to a hypersensitivity reaction in the context of bacterial or viral infection.23
On skin examination, patients most commonly present with 1 to 3 large (more than 10 mm), well-delimited lesions with a fibrinous necrotic center and symmetric distribution (Figure 4). Lesions are often described as “kissing.”22 Associated symptoms include a flulike illness or mononucleosislike syndrome.
This is a diagnosis of exclusion. A full review of systems should be obtained, and the differential should initially include other noninfectious causes of vulvar ulcers. Patients should be screened for underlying inflammatory bowel disease with fecal calprotectin.
Treatment depends on ulcer size. Small ulcers are treated with topical steroids, whereas larger lesions warrant a short course of oral prednisone until pain improves. Of note, systemic corticosteroids have not been shown to reduce disease duration.22 Pain control is another important component of treatment. Burning pain can be improved with copious amounts of zinc paste; patients also can be instructed to urinate while in a tub of water. Ulcers tend to resolve within 3 weeks without recurrence. Patient education should emphasize the natural course of disease and the fact that disease is not sexually transmitted.22
Pyoderma gangrenosum
Pyoderma gangrenosum (PG) is an idiopathic, inflammatory, ulcerative neutrophilic dermatosis that most commonly occurs on the lower extremities. The average age of onset is 40 to 60 years, and females are more commonly affected than males.24 Pyoderma gangrenosum is often associated with systemic disease such as inflammatory bowel disease, arthritis, malignancy, and hematological disease.25 Disease pathogenesis is unclear.
Pyoderma gangrenosum is a diagnosis of exclusion aided by evaluation of ulcer morphology, histopathology, and rapid response to systemic corticosteroids.26 Other entities important to exclude include infectious disease, Behçet disease, metastatic Crohn disease, and pemphigus vegetans.27 Pyoderma gangrenosum most commonly presents with 1 or more painful ulcers with violaceous, undermined borders. Most lesions affect the lower extremities or peristomal areas but may involve the upper extremities or genitalia, including the vulva. Genital PG is rare, but recognition is important. It should be considered in cases of genital ulcerations that are nonhealing and painful.26 Genital PG may be misdiagnosed as malignancy, leading to unnecessary radical surgery with associated morbidity and mortality. Studies has shown an inconsistent association between genital PG and systemic diseases.27,28
Treatment includes topical or systemic corticosteroids, minocycline, tacrolimus, cyclosporine, dapsone (Aczone), or surgical resection.26,29,30 With treatment, more than two-thirds of patients have been shown to achieve complete remission within 6 months, and 95% were in remission within 3 years.31
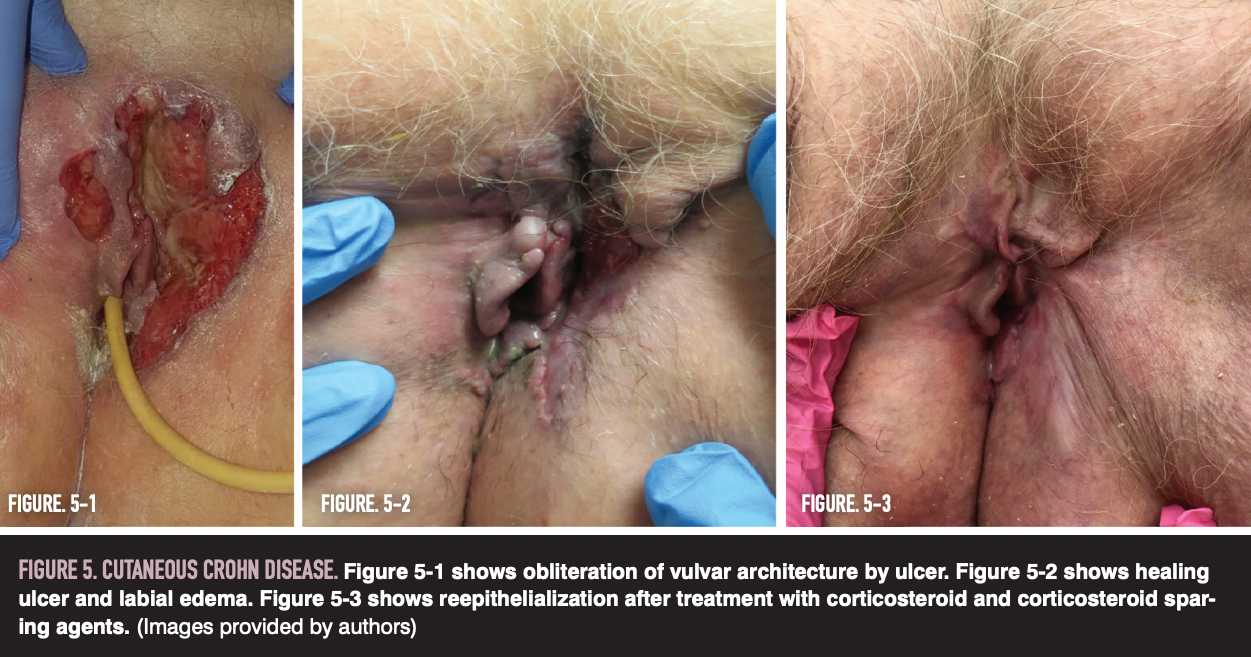
Cutaneous Crohn disease
Crohn disease (CD) is a relapsing-remitting full-thickness inflammatory disease of the gastrointestinal (GI) tract that can involve anywhere from the oral cavity to the anus. Crohn disease most often presents in early adulthood.32
Cardinal symptoms include abdominal pain, diarrhea, intermittent diarrhea, fatigue, and weight loss. Extraintestinal manifestations may involve musculoskeletal, ocular, skin, and hepatobiliary systems. Skin manifestations occur in up to one-third of patients and may be divided on an etiopathological basis into disease-specific lesions that share histopathology with underlying GI disease, reactive lesions secondary to inflammation, and associated conditions secondary to chronic inflammation or human leukocyte antigen linkages.33
Disease-specific lesions may involve the genital region and include perianal fissures and fistulae, and metastatic (cutaneous) lesions. Chronically, perianal fissures and fistulae may form abscesses and acrochordae, and form the majority of genital lesions in Crohn disease. Metastatic (cutaneous) lesions refer to inflammatory lesions involving areas not typically considered part of the underling Crohn disease. They can involve extremities, intertriginous areas, the face, and genitalia, and they are generally recognized as a rarer cutaneous manifestation of Crohn disease.33,34
Metastatic lesions involving the vulva are an underrecognized condition.35,36 Vulvoperineal Crohn disease (Figure 5) may occur in the absence of abdominal complaints, preceding gastrointestinal manifestations by months to years.33 Initial presentation may include vulvar or perineal pain.37 Physical exams can vary widely and may reveal vulvar ulcers in approximately half of vulvar cases, labial edema, or hypertrophic lesions.35,36
Definitive diagnosis requires biopsy because clinical appearance is not specific. Surgical outcomes are poor in cases of vulvoperineal Crohn disease, and treatment includes oral metronidazole, corticosteroid, or corticosteroid sparing agents such as azathioprine (Azasan) or infliximab.35 Some patients with vulvoperineal Crohn disease experience spontaneous healing, whereas others experience lesions that are refractory to medical therapy and require partial or total vulvectomy.38-40
Key takeaways
- Patients with pustular psoriasis of pregnancypresent with symmetric erythematous plaques with overlying sterile pustules at plaque margins; prompt treatment is essential.
- Hidradenitis suppurativa may affect the vulva. Patient support is available through the HS Foundation at www.hs-foundation.org.
- Acute vulvar aphthous ulcersare larger than oral aphthous ulcers and often require systemic steroids to improve symptoms.
- Pyoderma gangrenosum most commonly presents with 1 or more painful ulcers with violaceous, undermined borders. Genital pyoderma gangrenosum should be considered in cases of genital ulcerations that are nonhealing and painful.
- Vulvoperineal Crohn disease may occur in the absence of abdominal complaints, preceding gastrointestinal manifestations by months to years.
References
1. Trivedi MK, Vaughn AR, Murase JE. Pustular psoriasis of pregnancy: current perspectives. Int J Womens Health. 2018;10:109-115. doi:10.2147/IJWH.S125784
2. Oumeish OY, Farraj SE, Bataineh AS. Some aspects of impetigo herpetiformis. Arch Dermatol. 1982;118(2):103-105.
3. Flynn A, Burke N, Byrne B, Gleeson N, Wynne B, Barnes L. Two case reports of generalized pustular psoriasis of pregnancy: different outcomes. Obstet Med. 2016;9(2):55-59. doi:10.1177/1753495X15626623
4. Shaw CJ, Wu P, Sriemevan A. First trimester impetigo herpetiformis in multiparous female successfully treated with oral cyclosporine. BMJ Case Rep. 2011;2011:bcr0220113915. doi:10.1136/bcr.02.2011.3915
5. Oumeish OY, Parish JL. Impetigo herpetiformis. Clin Dermatol. 2006;24(2):101-104. doi:10.1016/j.clindermatol.2005.10.009
6. Shah A, Makhecha M. Pustular psoriasis of pregnancy with acrodermatitis continua of hallopeau. Indian J Dermatol. 2016;61(1):123. doi:10.4103/0019-5154.174149
7. Garg A, Lavian J, Lin G, Strunk A, Alloo A. Incidence of hidradenitis suppurativa in the United States: a sex- and age-adjusted population analysis. J Am Acad Dermatol. 2017;77(1):118-122. doi:10.1016/j.jaad.2017.02.005
8. Canoui-Poitrine F, Le Thuaut A, Revuz JE, et al. Identification of three hidradenitis suppurativa phenotypes: latent class analysis of a cross-sectional study. J Invest Dermatol. 2013;133(6):1506-1511. doi:10.1038/jid.2012.472
9. Liy-Wong C, Kim M, Kirkorian AY, et al. Hidradenitis suppurativa in the pediatric population: an international, multicenter, retrospective, cross-sectional study of 481 pediatric patients. JAMA Dermatol. 2021;157(4):385-391. doi:10.1001/jamadermatol.2020.5435
10. Vazquez BG, Alikhan A, Weaver AL, Wetter DA, Davis MD. Incidence of hidradenitis suppurativa and associated factors: a population-based study of Olmsted County, Minnesota. J Invest Dermatol. 2013;133(1):97-103. doi:10.1038/jid.2012.255
11. Revuz J. Hidradenitis suppurativa. J Eur Acad Dermatol Venereol. 2009;23(9):985-998. doi:10.1111/j.1468-3083.2009.03356.x
12. Lopez-Llunell C, Romaní J, Garbayo-Salmons P, Agut-Busquet E. Vulvar hidradenitis suppurativa: clinical cross-sectional study of 25 patients. J Dermatol. 2021;48(4):457-463. doi:10.1111/1346-8138.15728
13. Rastogi S, Patel KR, Singam V, et al. Vulvar cancer association with groin hidradenitis suppurativa: a large, urban, midwestern US patient population study. J Am Acad Dermatol. 2019;80(3):808-810. doi:10.1016/j.jaad.2018.10.008
14. Janse IC, Deckers IE, van der Maten AD, et al. Sexual health and quality of life are impaired in hidradenitis suppurativa: a multicentre cross-sectional study. Br J Dermatol. 2017;176(4):1042-1047. doi:10.1111/bjd.14975
15. Bernstein DI, Bellamy AR, Hook EW 3rd, et al. Epidemiology, clinical presentation, and antibody response to primary infection with herpes simplex virus type 1 and type 2 in young women. Clin Infect Dis. 2013;56(3):344-351. doi:10.1093/cid/cis891
16. Schillinger JA, McKinney CM, Garg R, et al. Seroprevalence of herpes simplex virus type 2 and characteristics associated with undiagnosed infection: New York City, 2004. Sex Transm Dis. 2008;35(6):599-606. doi:10.1097/OLQ.0b013e3181666fb1
17. Corey L, Spear PG. Infections with herpes simplex viruses (1). N Engl J Med. 1986;314(11):686-691. doi:10.1056/NEJM198603133141105
18. Corey L, Adams HG, Brown ZA, Holmes KK. Genital herpes simplex virus infections: clinical manifestations, course, and complications. Ann Intern Med. 1983;98(6):958-972. doi:10.7326/0003-4819-98-6-958
19. Kimberlin DW, Rouse DJ. Clinical practice. Genital herpes. N Engl J Med. 2004;350(19):1970-1977. doi:10.1056/NEJMcp023065
20. Benedetti J, Corey L, Ashley R. Recurrence rates in genital herpes after symptomatic first-episode infection. Ann Intern Med. 1994;121(11):847-854. doi:10.7326/0003-4819-121-11-199412010-00004
21. Workowski KA, Bachmann LH, Chan PA, et al. Sexually Transmitted Infections Treatment Guidelines, 2021. MMWR Recomm Rep. 2021;70(4):1-187. doi:10.15585/mmwr.rr7004a1
22. Vismara SA, Lava SAG, Kottanattu L, et al. Lipschütz’s acute vulvar ulcer: a systematic review. Eur J Pediatr. 2020;179(10):1559-1567. doi:10.1007/s00431-020-03647-y
23. Huppert JS. Lipschutz ulcers: evaluation and management of acute genital ulcers in women. Dermatol Ther. 2010;23(5):533-540. doi:10.1111/j.1529-8019.2010.01356.x
24. Binus AM, Qureshi AA, Li VW, Winterfield LS. Pyoderma gangrenosum: a retrospective review of patient characteristics, comorbidities and therapy in 103 patients. Br J Dermatol. 2011;165(6):1244-1250. doi:10.1111/j.1365-2133.2011.10565.x
25. Ashchyan HJ, Butler DC, Nelson CA, et al. The association of age with clinical presentation and comorbidities of pyoderma gangrenosum. JAMA Dermatol. 2018;154(4):409-413. doi:10.1001/jamadermatol.2017.5978
26. Sadowsky LM, Clark MA, Schlosser BJ. Pyoderma gangrenosum of the vulva treated with mycophenolate mofetil and infliximab. Dermatol Ther. 2019;32(3):e12905. doi:10.1111/dth.12905
27. Satoh M, Yamamoto T. Genital pyoderma gangrenosum: report of two cases and published work review of Japanese cases. J Dermatol. 2013;40(10):840-843. doi:10.1111/1346-8138.12252
28. Sau M, Hill NC. Pyoderma gangrenosum of the vulva. BJOG. 2001;108(11):1197-1198. doi:10.1111/j.1471-0528.2003.00261.x
29. Chen JR, Chen SS, Chan YJ. Rapid recovery of vulvar pyoderma gangrenosum in response to aggressive surgery and steroid treatment. Taiwan J Obstet Gynecol. 2014;53(1):97-100. doi:10.1016/j.tjog.2012.12.006
30. Maloney C, Blickenstaff N, Kugasia A, Buford LB, Hoffman MD. Vulvovaginal pyoderma gangrenosum in association with rituximab. JAAD Case Rep. 2018;4(9):907-909. doi:10.1016/j.jdcr.2018.09.001
31. Bennett ML, Jackson JM, Jorizzo JL, Fleischer AB Jr, White WL, Callen JP. Pyoderma gangrenosum. A comparison of typical and atypical forms with an emphasis on time to remission. Case review of 86 patients from 2 institutions. Medicine (Baltimore). 2000;79(1):37-46. doi:10.1097/00005792-200001000-00004
32. Loftus EV, Jr. Clinical epidemiology of inflammatory bowel disease: incidence, prevalence, and environmental influences. Gastroenterology. 2004;126(6):1504-1517. doi:10.1053/j.gastro.2004.01.063
33. Hagen JW, Swoger JM, Grandinetti LM. Cutaneous manifestations of Crohn disease. Dermatol Clin. 2015;33(3):417-431. doi:10.1016/j.det.2015.03.007
34. Peltz S, Vestey JP, Ferguson A, Hunter JA, McLaren K. Disseminated metastatic cutaneous Crohn’s disease. Clin Exp Dermatol. 1993;18(1):55-59. doi:10.1111/j.1365-2230.1993.tb00969.x
35. Barret M, de Parades V, Battistella M, Sokol H, Lemarchand N, Marteau P. Crohn’s disease of the vulva. J Crohns Colitis. 2014;8(7):563-570. doi:10.1016/j.crohns.2013.10.009
36. Foo WC, Papalas JA, Robboy SJ, Selim MA. Vulvar manifestations of Crohn’s disease. Am J Dermatopathol. 2011;33(6):588-593. doi:10.1097/DAD.0b013e31820a2635
37. Leu S, Sun PK, Collyer J, et al. Clinical spectrum of vulva metastatic Crohn’s disease. Dig Dis Sci. 2009;54(7):1565-1571. doi:10.1007/s10620-008-0448-y
38. Baker VV, Walton LA. Crohn’s disease of the vulva. South Med J. 1988;81(2):285-286. doi:10.1097/00007611-198802000-00038
39. Fenniche S, Mokni M, Haouet S, Ben Osman A. [Vulvar Crohn disease: 3 cases]. Ann Dermatol Venereol. 1997;124(9):629-632.
40. Shen RN, Cybulska BA, Thin RN, McKee PH. Vulval Crohn’s disease mimicking genital herpes. Int J STD AIDS. 1993;4(1):54-56. doi:10.1177/095646249300400112

Newsletter
Get the latest clinical updates, case studies, and expert commentary in obstetric and gynecologic care. Sign up now to stay informed.
A legacy of excellence: Reflecting on the final print edition of Contemporary OB/GYN
April 25th 2025Marking the end of an era and the beginning of a new chapter, the final print edition of Contemporary OB/GYN celebrates over 50 years of evidence-based guidance and unwavering support for clinicians.
Read More
