On The Pulse

Child protective services (CPS) is supposed to protect against maltreatment for adolescents, but do they help protect against negative outcomes linked to sexual health?

In this episode of Pap Talk, Gloria Bachmann, MD, MSc, breaks down what it means to be a health care provider for incarcerated individuals, and explores the specific challenges women and their providers face during and after incarceration. Joined by sexual health expert Michael Krychman, MD, Bachmann also discusses trauma-informed care and how providers can get informed.

Agile Therapeutics, Inc. recently announced a new alliance with Afaxys Pharma, LLC to promote levonorgestrel and ethinyl estradiol (Twirla) transdermal system.

The findings support previous research demonstrating that increased access to emergency contraception does not compromise use of other more effective methods or lead to an increase in reported levels of unprotected sexual intercourse, according to Hawkins.

Many adolescents across the United States lack quality sex education, a trend that has worsened in recent years. Moreover, national statistics mask significant inequities in the receipt of formal sex education by gender, race and ethnicity, sexual orientation, and the location of instruction, according to a study in the Journal of Adolescent Health.

Consensus group discussion resulted in a shift from results-based to risk-based management for cervical cancer screening.

While it has been nearly 50 years since vaginal dilators were introduced as a therapy to decrease VS, the medical community still knows very little about the potential impact on long term outcomes.
.jpeg?fit=crop&auto=format)
A population-based retrospective cohort study in JAMA Network Open concluded that maternal sexually transmitted infections (STIs) are significantly linked to an increased risk of preterm birth in the United States.
It is essential to expand access to all available, effective cervical cancer screening options for the most vulnerable populations. The targeted use of any particular screening modality in higher-risk populations is not supported by evidence-based guidelines; any effective screening is the priority.
Surviving a bout of cancer early in life puts children and teenagers at risk of cancers linked to human papillomavirus (HPV). A phase 2 trial examined how effective a 3-dose series was in providing protection.
State legislation that allows adolescents to choose to receive the HPV vaccine without parental interference would elevate immunization rates, according to a new study.

Telehealth offers a whole new way to reach out to adolescent patients for critical conversations on sexual health, according to a session at the virtual 2021 American Academy of Pediatrics National Conference & Exhibition.

This episode features Scott Kober, Michael Krychman, MD, and James Keller, MD, as they discuss the changes and paradigms of health care delivery.
Not only are chlamydia and gonorrhea 2 of the most common sexually transmitted infections (STIs) in the United States, but people are being diagnosed with them at a higher rate than ever before, according to the Centers for Disease Control and Prevention (CDC).

Associate Editor Lindsey Carr sat down with Gary Schoolnik, MD, Visby Medical’s Chief Medical Officer, to learn more about the device’s functionality and application.

The U.S. Food and Drug Administration (FDA) has issued 501(k) clearance for Visby Medical’s Sexual Health Click Test, a single-use, instrument-free polymerase chain reaction (PCR) diagnostic test for sexually transmitted infections (STI).

The COVID-19 pandemic has affected the global community in countless ways, some of which are more obvious than others.

Featuring Dr. Krychman and Nurse Barb Dehn.

The data suggest that both SCD providers and AYA need better evidence and educational resources for contraception, as well as coordinated SRH counseling, according to researchers.
The CDC updated its guidelines for diagnosing and treating STIs, which included updates on the diagnosis and treatment of bacterial vaginosis (BV) in pregnant and nonpregnant women.

The Centers for Disease Control and Prevention (CDC) updated its guidelines for the treatment of sexually transmitted infections (STIs), notably for Neisseria gonorrhoeae, Chlamydia trachomatis, Trichomonas vaginalis and pelvic inflammatory disease (PID).

The management of diabetes is multifaceted, often requiring the collaboration of a multidisciplinary team of providers. While sexual side effects of diabetes are not commonly discussed during patient visits, there is a correlation between the presence of diabetes and frequent sexual dysfunction in women.
For cervical cancer screening, human papillomavirus (HPV)–based screening modalities detect significantly more abnormal cervical cells than traditional liquid-based cytology (LBC) approaches, according to a study in JAMA Network Open.

There are many reasons why a caregiver may refuse to have his or her child receive the human papillomavirus (HPV) vaccine, including safety concerns. A research letter examines whether those concerns have increased.

“Over the past several years, a growing number of companies have been providing contraception through online platforms, but there has been very little research on these businesses,” said principal investigator Brittni Frederiksen, MPH, PhD, a senior policy analyst at KFF in San Francisco.

To gauge progress that had been made in the decade since the establishment of CREOG, more than 4,000 U.S.-based ob-gyn residents were sent a 32-item survey in June 2016 to gauge their comfort level in asking patients about their sexual history and providing counseling to patients of varying ages and ethnic/racial backgrounds.

The management of diabetes is multifaceted, often requiring the collaboration of a multidisciplinary team of providers. While sexual side effects of diabetes are not commonly discussed during patient visits, there is a correlation between the presence of diabetes and frequent sexual dysfunction in women.
The authors of the prospective study in the journal Health Technology Assessment believe the simple intervention has enormous potential to prevent more unintended pregnancies after emergency contraception.

The interaction of sexual orientation and provider-patient communication on sexual and reproductive health (SHR) can be tricky to navigate, according to the results of a prospective study of a sample of U.S. women of diverse sexual orientations in the journal Patient Education and Counseling.

Angie DeRosa, Senior Editor at Contemporary OB/GYN®, interviewed Michael Krychman, MD, about the importance of creating an affirming environment in medical facilities for the LGBTQ+ community. He is the executive director of the Southern California Center for Sexual Health and Survivorship in Newport Beach, Calif.

A webinar held by the Kaiser Family Foundation (KFF) in April shared highlights from the 2020 Women’s Health Survey, followed by a panel discussion on its implications in several areas. This article captures the discussion portion of the webinar.

In this video, Angie DeRosa, senior editor of Contemporary OB/GYN®, interviews Michael Krychman, MD, a gynecologist who specializes in sexual health and survivorship medicine.

Poor sleep quality is linked to female sexual dysfunction, whereas good sleep quality is associated with heightened sexual activity, according to a cross-sectional analysis of the Data Registry on Experiences of Aging, Menopause and Sexuality (DREAMS).

Limited changes occur in endocrine markers with the combined oral contraceptive Nextstellis (Mayne Pharma), according to a prospective study presented at the virtual annual meeting of the International Society for the Study of Women's Sexual Health (ISSWSH) in March.

A weekend workshop with a group therapy intervention program promoted safe sex and reduced drinking.

The vast majority of cancer survivors reported that they experience sexual side effects after chemotherapy, radiation and other cancer treatments according to the results of a survey presented at the annual meeting of the American Society for Radiation Oncology.

Dr. Krychman is a gynecologist who specializes in sexual health and survivorship medicine. He is executive director of the Southern California Center for Sexual Health and Survivorship Medicine Inc. in Newport Beach, Calif.
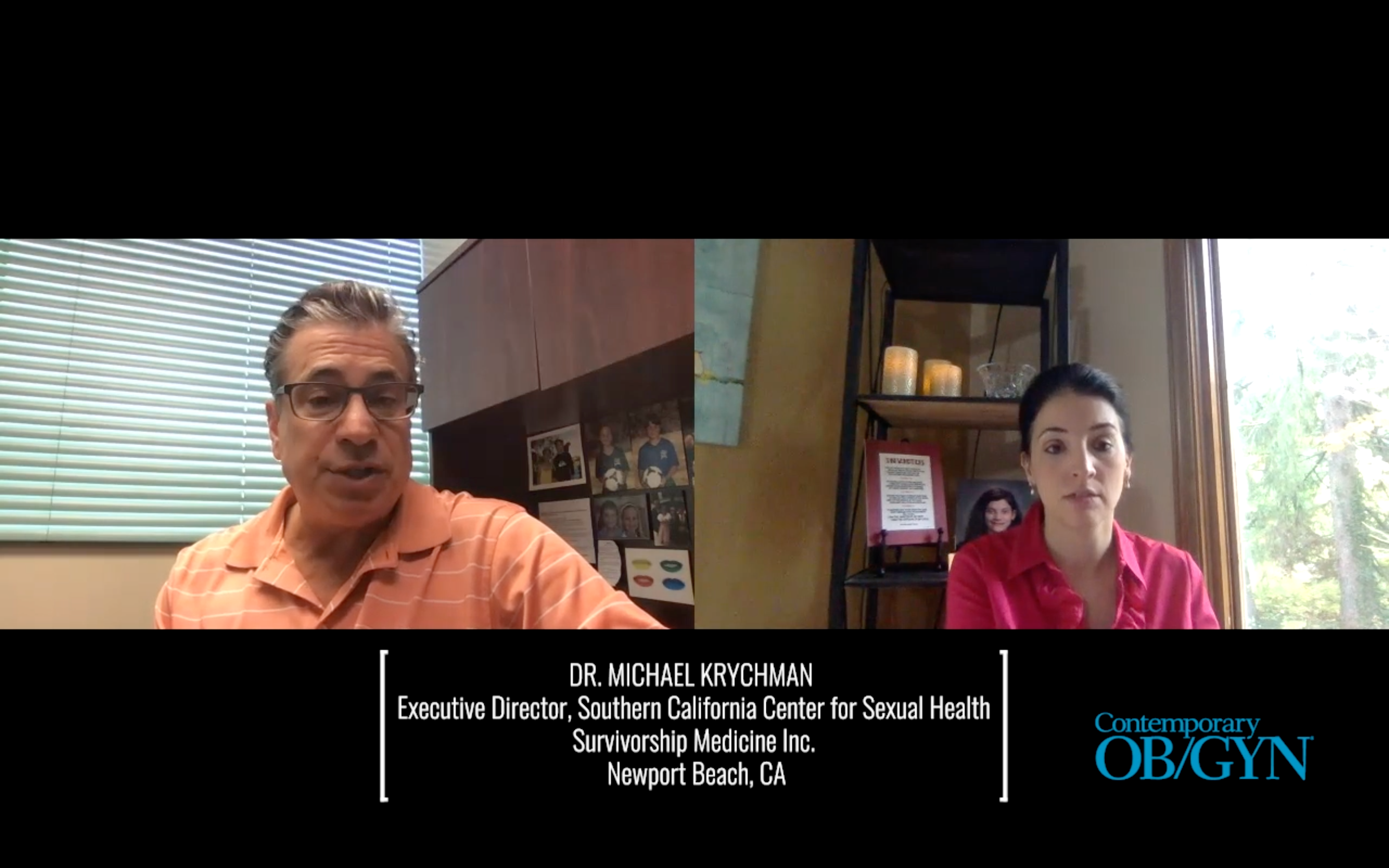
Dr. Krychman is a gynecologist who specializes in sexual health and survivorship medicine. He is executive director of the Southern California Center for Sexual Health and Survivorship Medicine Inc. in Newport Beach, Calif.

Patients and practitioners may be hesitant to delve into sexual health, but talking about it is one way to normalize it.